Chronic pelvic pain and brain-gut axis: what is the involvement of the gut microbiota?
By Claire Cardaillac1,2 Martial Caillaud 2 Michel Neunlist 2
1 Department of Gynaecology-Obstetrics and Reproductive Medicine, Nantes University Hospital, Nantes, France
2 University of Nantes, Inserm, TENS, The Enteric Nervous System in Gut and Brain Diseases, IMAD, Nantes, France
Lay public section
Find here your dedicated section
Sources
This article is based on scientific information
Sections
About this article
Chronic pelvic pain is common, debilitating and particularly affects women. It can be caused by identified lesions, such as endometriosis, or functional syndromes characterised by visceral hypersensitivity, such as irritable bowel syndrome (IBS) or interstitial cystitis. In such cases, peripheral and central sensitisation mechanisms trigger a lowering of pain thresholds and lead to diffuse and difficult-to-treat symptoms.
The gut microbiota plays an increasingly important role in understanding this type of pain. Through its interactions with the immune and nervous systems and metabolism, it exerts a direct effect on the excitability of sensory fibres and pain circuits. Some bacterial metabolites favour neuronal inflammation and hyperexcitability, while others exert a protective effect via anti-inflammatory mediators or endogenous opioids.
In IBS, dysbiosis characterised by a loss of beneficial bacteria (e.g. Faecalibacterium, Roseburia) and increase of opportunistic bacteria has been documented, with experimental evidence of its causal role. In endometriosis, the gut microbiota may contribute to lesion progression and oestrogen modulation, suggesting a bidirectional interaction between the gut microbiota and disease.
These findings are paving the way for new therapeutic avenues including probiotics, prebiotics, postbiotics and even faecal-microbiota
transplantation. Although data remain preliminary, targeting the microbiota appears to offer a promising strategy for improving the management of chronic pelvic pain.
From nociperception to chronic pelvic pain
Pain is defined as an unpleasant sensory and emotional experience, which may or may not be associated with tissue damage. It is referred to as chronic when the pain persists for over three months, resists treatment and leads to changes in functional abilities and social relationships. Chronic visceral pelvic pain is characterised by deep, throbbing and diffuse pain, making its diagnosis complex and imprecise. This type of pain is particularly common in women and may require input from several internal-medicine specialists, i.e. gastroenterology, gynaecology and urology.
Pelvic pain can be caused by several types of organ lesions (e.g. endometriosis) or functional syndromes characterised by visceral hypersensitivity (e.g. irritable bowel syndrome (IBS) or interstitial cystitis). A combination of several painful pelvix condition is often observed in a single patient. Other patients may experience debilitating pain for which no specific cause has been identified. Internal-medicine specialists usually analyse pain as the expression of a single lesion. While it is certainly necessary to treat the injured organ, this may sometimes not be enough to provide relief to patients with chronic pelvic pain. Some patients present with an extensive set of symptoms, combining sensitivity disorders with issues affecting several pelvic organ functions at the same time. Such phenomena are linked to sensitisation mechanisms that appear several months or years after the onset of pain 1. This type of sensitisation is characterised by a lowering of sensitivity thresholds leading to increased pain or pain provoked by stimuli of normally non-nociceptive intensity (e.g. intolerance to the sensation of rectal filling). Pain spreading over time has also been described. In fact, the painful sensation can persist despite the lack
of stimulation (e.g. post-defecation pain). Finally, pain spreading beyond the stimulated area is also observed (e.g. pain during rectal filling causing bladder pain).
On a pathophysiological level, a distinction is made between psychological, adaptive and chronic pain. Acute pain occurs following an initial painful stimulus (heat, pressure, pH variation or algogenic substances) which activates receptors at the endings of the peripheral nociceptive fibres. In the case of the viscera, sensory innervation occurs in the spinal nerves whose endings are located in the muscles and/or mucosa of organs with cell bodies in the dorsal root ganglia 2. Under physiological conditions, activation of Aδ or C afferent fibre receptors generates an action potential (AP) that is translated into nociceptive information in the dorsal root ganglia. This information is then transmitted to the second-order spinal neurons in the dorsal horn. Second-order neurons transmit information to the thalamus via the spinothalamic and spinoreticulothalamic pathways. In the thalamus, the third neuron transmits the message to various areas in the cortex (prefrontal, cingulate,
somatosensory or insular cortex). This third neuron is responsible for transforming nociception into pain, with its affective-emotional, sensitive, cognitive and behavioural dimensions.
In the case of chronic visceral pain observed in conditions such as IBS or chronic inflammatory bowel disease (IBD), particularly
in remission, hypersensitivity of intestinal nociceptive fibres has been documented 3.
This hypersensitivity may be partly due to gut permeability alterations, thereby increasing the passage of food or bacterial antigens, leading to inflammatory mechanisms with mast-cell recruitment and release of pro-inflammatory mediators such as histamine or proteases. This inflammatory environment could contribute to hyperexcitability in the nociceptive fibres, which in turn contribute to maintaining the inflammatory microenvironment by releasing neuropeptides (Substance P: SP and Calcitonin-Gene Related Peptide: CGRP). These repeated stimuli trigger phenotypic and excitability alterations to the nociceptive neurons in the Dorsal Root Ganglion (DRG), known as peripheral sensitisation. In the spine, repeated APs from DRG neurons increase the release of excitatory neurotransmitters such as glutamate 4. In the long term, this leads to synaptic reinforcement by increasing the number of glutamatergic receptors, combined with dysfunction of the inhibitory systems, resulting in sensitisation of the spinal neurons 4. Thus, central sensitisation is a pathological state of nociception function linked to its dysfunction, with a strengthening of facilitatory systems and lowering of pain-inhibitory systems.
The microbiota, a potential pain modulator
The factors responsible for visceral fibre hypersensitivity are still poorly understood, but the gut microbiota could play a role (figure 1).
Figure 1 : Mechanisms of abdominal pain
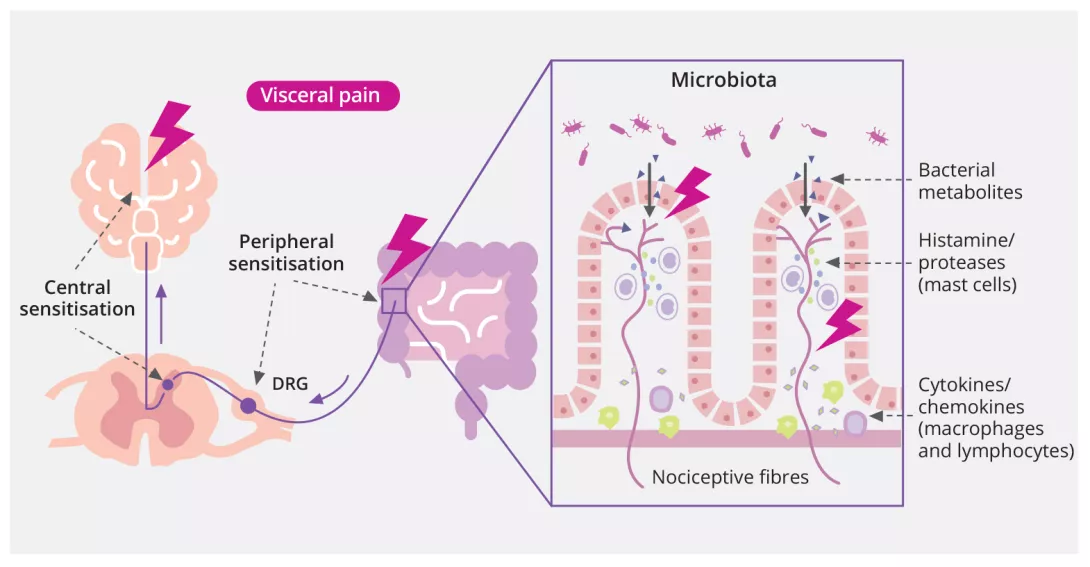
The microbiota is defined as all of the microorganisms living in a specific environment in a host. It is mainly composed of bacteria, but also includes viruses, yeasts and protozoa. These micro-organisms may be present without having any impact
on their host (commensalism) or they may interact closely with it.
The number of bacteria colonising the human body (3.8∙1013) is roughly the same as the number of human host cells in adulthood (3.0∙1013) 5. The gut microbiota plays a key role in the two-way communication between the gut and various organs, including the brain. For several years this has been referred to as the microbiota-gut-brain axis. In the past, studies have typically focused on the role of the microbiota in gastrointestinal disorders (irritable bowel syndrome, inflammatory bowel disease). More recently, it has been recognised that dysfunction of this axis is involved in the pathophysiology of many other conditions, including metabolic diseases (obesity, diabetes) and neurological diseases (autism, Parkinson’s disease, depression).
The gut microbiota and the brain communicate with each other using several pathways such as the vagal nervous system, the immune system and the humoral pathways, after modulation of enteroendocrine functions. Nociceptive afferent fibres can also be directly modulated by various bacterial metabolites 6. The main mediators identified are bacterial metabolites (e.g. short-chain fatty acids, secondary bile acids), neurotransmitters and neuromodulators (e.g.: GABA) and bacterial products (e.g.: PAMPs, tryptophan derivatives). Some molecules increase neuronal excitability by activating nociceptors, producing Nerve Growth Factor (NGF) and increasing local inflammation. Others,
however, have the opposite effect (e.g.: GABA) and can inhibit the transmission of the nociceptive message by producing endogenous opioids or anti-inflammatory mediators.
Clinical examples: irritable bowel syndrome and endometriosis
Irritable bowel syndrome
IBS is characterised by chronic functional intestinal disorders, mainly combining abdominal pain and bowel disorders (diarrhoea, constipation or both in alternation). This condition affects around 5-10% of the population, primarily young adult females. The pathophysiology of IBS is not yet fully understood but it has been extensively documented that gut-brain-communication changes lie at the root of digestive motility disorders and visceral hypersensitivity. On a central level, IBS patients experience impaired information processing,
hypervigilance and increased anxiety. In recent years, the gut microbiota has been proposed as one of the causative factors in IBS 7 and several studies have highlighted changes in the composition and diversity of the microbiota in IBS.
For instance, a systematic review showed that the phylum Firmicutes decreased and the phylum Bacteroidetes increased in patients with IBS and diarrhoea 8.
A reduction in the abundance of the genus Bifidobacterium was also found in stool and mucosal samples from IBS patients, along with an increase in the genus Bacteroides. An increase in pathogens (i.e. Escherichia coli and Enterobacterium families) was also observed. An enrichment of certain bacterial taxa such as Enterobacteriaceae, Streptococcus, Fusobacteria, Gemella and Rothia, and a depletion of bacterial genera recognised as beneficial to health, such as Roseburia and Faecalibacterium, were also observed in IBS patients. In IBS, the causal role of these kinds of microbiota changes has been strongly suggested in view of the ability to induce some symptoms in
preclinical models after the transfer of stools from IBS patients 9. In addition, the role of several mediators produced by the intestinal microbiota, such as LPS, short-chain fatty acids and secondary bile acids, has been suggested in chronic abdominal pain, visceral hypersensitivity and gut inflammation 10. The gut microbiota could therefore be a major cofactor in chronic abdominal pain and associated
inflammation.
Endometriosis
Endometriosis is defined as the transplantation of endometrial cells outside the uterine cavity, which may be facilitated by chronic pelvic inflammation. The main symptoms of endometriosis are chronic pelvic pain, gastrointestinal problems and infertility. A systematic review analysed studies of the gut microbiota in women with endometriosis and chronic pelvic pain 11. A total of 28 clinical studies and six animal studies were included in the review. In these human and animal studies, increased gut microbiota diversity was observed in the endometriosis groups. However, there was no clear consensus on the composition of the microbiota associated with endometriosis. None of the studies analysed composition or diversity based on the characteristics of pain.
Animal studies (6/6) confirmed the bidirectional relationship between the gut microbiota and the onset and progression of endometriosis. Endometriosis induction in mice actually induced changes in the gut microbiota.
In the study by Yuan, no early-stage differences were observed after endometriosis induction versus a control group. Differences appeared 21 days after the start of the experiment and increased thereafter, with decreased diversity and richness of the microbiota in the endometriosis group, increased genera Bifidobacterium, Proteobacteria and Verrucomicrobia, decreased Bacteroidetes and Firmicutes phyla and an increase in the Firmicutes/Bacteroidetes ratio, reported 12. Conversely, treatments targeting the microbiota, such as broad-spectrum antibiotics, reduced the volume and weight of endometriosis lesions, induced a decrease in endometrial
lesion cell proliferation and inflammatory markers (cytokines, macrophages) 13. Apart from inflammation, the gut microbiota could contribute to the pathophysiology of endometriosis through its role in regulating oestrogen metabolism. A specific microbiota called the estrobolome actually plays a central role in hormone regulation, especially oestrogen 11. Estrobolome contains bacteria that produce beta-glucuronidase,
which is an enzyme able to modify the active form of oestrogen. Gut microbiota disturbances could therefore lead to increased circulating oestrogen levels and favour the development of endometriosis. However, these studies did not investigate pain modulation.
Therapeutic prospects via the microbiota
The development of therapeutic approaches modulating the composition or function of the gut microbiota are becoming increasingly recognised as playing a complementary role in the current therapies used for the management of chronic functional digestive or pelvic conditions. The basic aim of these approaches is to restore a balanced and functional microbiota (via dietary interventions or prebiotics), or provide bacteria with beneficial effects for the host (probiotics). Specific combinations of probiotics, or specific species and strains 14, appear to have beneficial effects on overall IBS symptoms and abdominal pain [15]. Symbiotic approaches combining pre- and probiotics are also used. Approaches based on the transplantation of faecal microbiota also suggest a therapeutic potential in functional diseases such as IBS 16. In addition, an increasing number of studies are suggesting that the effectiveness of the response to faecal-microbiota transplantation in IBS or to probiotic treatments is influenced by the composition of the microbiota of the recipient.
In the case of endometriosis, two randomised clinical studies have suggested that probiotics were effective in improving pain 17, 18, although one study reported that the efficacy did not last after discontinuing the probiotics.
In general, the effectiveness of these approaches in the management of pelvic functional disorders, although demonstrated in some cases, remains limited by the small population sizes in the studies and, above all, by the wide diversity and variability of symptoms observed in these disorders, as shown in a recent literature review 15.

