Fecal transplant and recurrent Clostridium difficile infections: bacteriophages are necessary in donors
According to a Canadian study, the diversity and relative abundance in donors’ bacteriophages seem to have an impact on the success of fecal transplants in patients with recurrent Clostridium difficile infections.
Lay public section
Find here your dedicated section
Sources
This article is based on scientific information
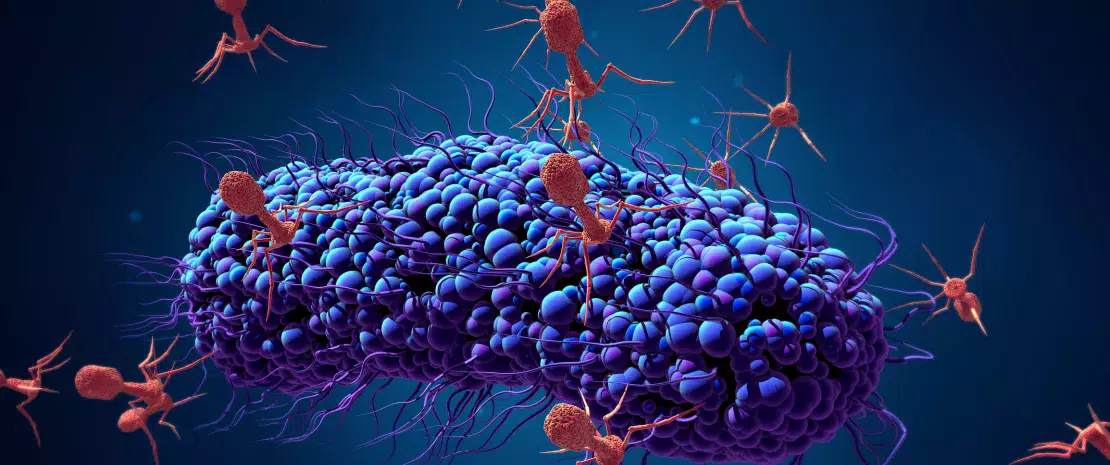
About this article
Despite the positive results obtained with fecal microbiota transplants (FMT) as a first-line treatment for recurrent Clostridium difficile infections (rCDI), 8 to 50% of patients relapse. Recent works have shown the potential influence of bacteriophages–a virus that infects bacteria within the gut microbiota–on the success rate of fecal transplants. A Canadian team thus studied the impact of bacteriophage populations in donors and patients with rCDI treated with FMT via colonoscopy.
Impact of FMT on bacteriophages
Unsurprisingly, all 19 recipients from this trial had a less diversified gut microbiota than the 7 donors. The diversity of their bacteriophages was greater than that of donors and of the 96 control subjects. This was potentially due the administration of vancomycin in the 24 hours before the transplant, which could trigger the induction of phages. The FMT, which was effective from the first administration in 12 patients, led to a reduction of this increased diversity. For the 7 patients who received a second FMT, bacteriophage diversity was only reduced on the second attempt.
Importance of donors’ profile
On the bacterial front, recipients’ profile became more similar to the donors’ after a successful transplant: increased levels of Bacteroidetes and Firmicutes, decreased levels of Proteobacteria, which were prevalent until then (mainly species from the Klebsiella and Escherichia genera). Nevertheless, no difference was observed between patients who had a positive response after the first transplant and those who did not. On the contrary, donors’ bacteriophages seem to be correlated to the successful or failed outcome of the transplant: greater diversity as well as decreased relative abundance were associated to a successful FMT. But before using donors’ bacteriophage composition as a prognostic factor for FMT in the treatment of rCDI, the researchers call for revised research methods into this facet of the gut flora which is still largely unknown. They believe these results are probably only the “tip of the iceberg”.
Explain fecal transplantation to your patients with this dedicated content: