Functional gastrointestinal disorders
Functional gastrointestinal disorders (FGDs), the most common intestinal afflictions, are a collection of chronic digestive symptoms that are not explained by any detectable anatomic anomaly.
- Learn all about microbiota
- Microbiota and related conditions
- Act on your microbiota
- Publications
- About the Institute
Healthcare professionals section
Find here your dedicated section
Sources
This article is based on scientific information
Sections

About this article
Irritable bowel syndrome (IBS), the most common FGD
FGDs encompass a set of symptoms such as IBS, constipation, diarrhea, functional bloating, and non-specific FGDs.
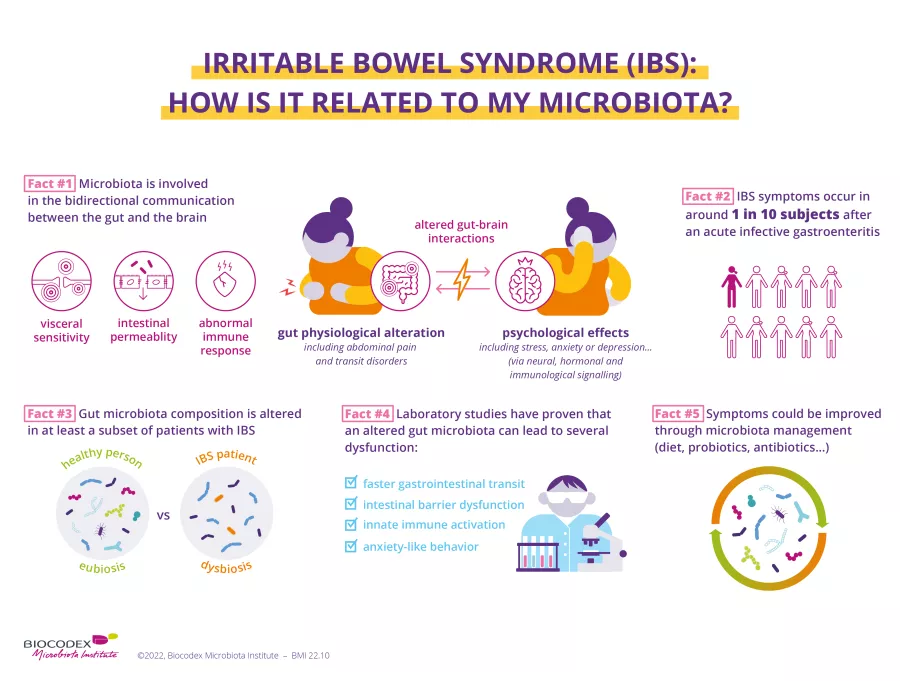
IBS alone affects 10% of the population and is distinguished from other FGDs by abdominal pain associated with constipation, diarrhea, or alternations between them. It often presents with abdominal bloating and a higher level of stress than the general population.
52% Just 1 in 2 people having suffered from a digestive condition involving the microbiota had made the connection
Irritable Bowel Syndrome
Irritable Bowel Syndrome (IBS) is a functional gastrointestinal disorder characterized by recurrent abdominal pain, that is associated with changes in stool frequency or stool form, in the absence of any organic disorder. Using ROME IV criteria, IBS is classified into four subtypes:
- IBS with predominant constipation (IBS-C),
- IBS with predominant diarrhea (IBS-D),
- IBS with mixed bowel habits (IBS-M),
- IBS, unsubtyped (IBS-U) which does not meet the criteria for IBS-C, D, or M
Psychiatric comorbidities, such as anxiety, depression and somatization are common in patients with IBS.
Irritable bowel syndrome (IBS), previously called a “functional bowel disorder”, is the most common gut-brain axis disorder.
FGDs don’t spare children
In very small children, FGDs represent the most common gastrointestinal reason for a doctor’s visit. This includes baby colic with regurgitation and constipation problems, IBS, and other less well characterized functional problems. Stomach pain, bloating, diarrhea, and constipation are commonly associated with FGDs and can have major consequences on a child’s daily life. Stress and anxiety can also favor or prolong certain symptoms, particularly pain.
Disrupted communication between the intestines and the brain
The causes of IBS are still poorly understood. The risk of developing IBS increases five-fold after a bacterial infection causing acute diarrhea. It has been suggested that it could be related to dysfunction in the communication between the brain and the intestine, in conjunction with an imbalance in the intestinal flora. In the majority of cases, there is a (sidenote: Dysbiosis Generally defined as an alteration in the composition and function of the microbiota caused by a combination of environmental and individual-specific factors. Levy M, Kolodziejczyk AA, Thaiss CA, et al. Dysbiosis and the immune system. Nat Rev Immunol. 2017;17(4):219-232. ) among the bacterial species that make up the microbiota, with fewer positive bacteria and more harmful bacteria. This dysfunction causes intestinal motor problems: transit is slowed, the intestinal barrier is modified, and slight inflammation develops. It also causes hypersensitivity in the mucosa that makes normal phenomena, like the movement of intestinal gas, painful.
Have you heard of "dysbiosis"?
It refers to a breakdown in the delicate balance between the billions of microorganisms that make up our microbiota and in their relationship with our body.
Promising data for probiotics
For adults, in addition to a controlled diet, treatment options include antispasmodics, laxatives, and antidiarrheals. In children, relaxation and hypnosis techniques, which can relieve pain, are preferred. Sometimes antispasmodics are also prescribed. To modify the microbiota, there are promising data currently available about probiotics, particularly
(sidenote:
Bifidobacterium
A genus of Y-shaped bacteria, most species of which are beneficial to humans. They are found in the gut of humans, and in some yogurts.
They:
- Protect the gut barrier
- Participate in the development of the immune system and help fight inflammation
- Promote digestion and improve symptoms of gastrointestinal disorders
Sung V, D'Amico F, Cabana MD, et al. Lactobacillus reuteri to Treat Infant Colic: A Meta-analysis. Pediatrics. 2018 Jan;141(1):e20171811.
O'Callaghan A, van Sinderen D. Bifidobacteria and Their Role as Members of the Human Gut Microbiota. Front Microbiol. 2016 Jun 15;7:925.
Ruiz L, Delgado S, Ruas-Madiedo P, et al. Bifidobacteria and Their Molecular Communication with the Immune System. Front Microbiol. 2017 Dec 4;8:2345.
)
and
(sidenote:
Lactobacilli
Rod-shaped bacteria whose main characteristic is the production of lactic acid, from where they get the name “lactic acid bacteria”.
Lactobacilli are present in the oral, vaginal and gut microbiota of humans, but also in plants and animals. They are found in fermented foods, such as dairy products (e.g. certain cheeses and yoghurts), pickles, sauerkraut, etc.
Lactobacilli are also found in probiotics, with certain species recognized for their beneficial properties.
W. H. Holzapfel et B. J. Wood, The Genera of Lactic Acid Bacteria, 2, Springer-Verlag, 1st ed. 1995 (2012), 411 p. « The genus Lactobacillus par W. P. Hammes, R. F. Vogel
Tannock GW. A special fondness for lactobacilli. Appl Environ Microbiol. 2004 Jun;70(6):3189-94.
Smith TJ, Rigassio-Radler D, Denmark R, et al. Effect of Lactobacillus rhamnosus LGG® and Bifidobacterium animalis ssp. lactis BB-12® on health-related quality of life in college students affected by upper respiratory infections. Br J Nutr. 2013 Jun;109(11):1999-2007.
)
, and about fecal transplants. However, large-scale clinical trials still have to be conducted to confirm each of these options.
The gut microbiota
- Koloski, NA, Talley, NJ, and Boyce, PM. Epidemiology and health care seeking in the functional GI disorders: a population-based study. Am J Gastroenterol. 2002 ; 97, 2290-2299.
- Chouraqui JP, Thomassin N. Douleurs abdominales récurrentes, syndrome de l’intestin irritable ou dyspepsia chez l’enfant. Archives de pédiatrie 2009; 16: 855-7.
- Spiller R, Garsed K. Postinfectious irritable bowel syndrome. Gastroenterology 2009; 136: 1979-88.
- Collins SM, Surette M, Bercik P. The interplay between the intestinal microbiota and the brain. Nat Rev Gastroenterol Hepatol 2012; 10: 735-42.
- Simren M, Barbara G, Flint HJ et al. Intestinal microbiota in functional bowel disorders: a Rome foundation report. Gut 2013; 62: 159-76.
- Camilleri M. Peripheral mechanisms irritable bowel syndrome. N Engl J Med 2012; 367: 1626-35.
- Jean-Marc Sabaté JM, Pauline Jouët. Prise en charge du Syndrome de l’Intestin Irritable (SII), septembre 2016. Société Nationale Française de Gastro-Entérologie.
- Ford AC et al. Efficacy of prebiotics, probiotics and synbiotics in irritable bowel syndromeand chronic idiopathic constipation: systematic review and meta-analysis. Am J Gastroenterol 2014; 109: 1547-61.
- Giglione E et al. The association of Bifidobacterium breve BR03 and B632 is effective to prevent colics in bottle-fed infants: a pilot, controlled, randomized, and double-blind study. J Clin Gastroenterol 2016; 50 suppl2: S164-S167.
- Moayyedi P, Ford AC, Talley NJ et al. The efficacy of probiotics in the treatment of irritable bowel syndrome. A systematic review. Gut 2010; 59: 325-32.
- Rossen NG, MacDonald JK, de Vries EM. Fecal microbiota transplantation as novel therapy in gastroenterology: a systematic review. World J gastroenterol 2015; 21: 5359-71.