Antibiotics: Dr Jekyll and Mr Hyde
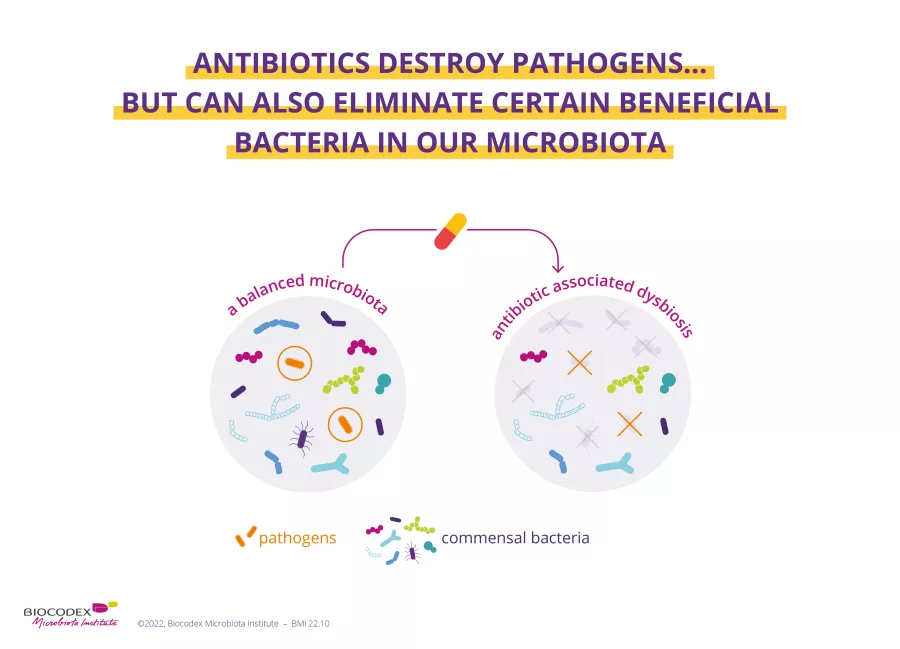
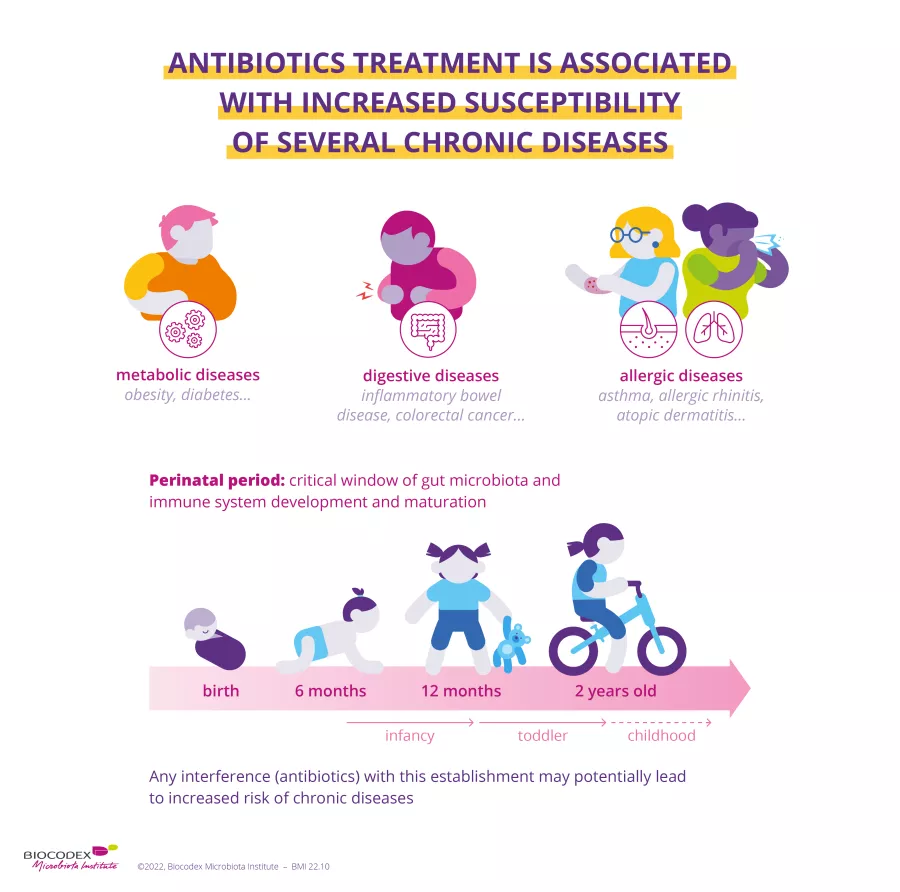
Undoubtedly one of the most important scientific discoveries of the 20th century, antibiotics save millions of lives each year. However, their effectiveness is now under threat from the appearance of multiple drug resistance. By destroying the bacteria responsible for infection, antibiotics can also lead to dysbiosis. Spotlight on a therapeutic weapon to be handled with care.

 Antimicrobial resistance: a global threat, an international response
Antimicrobial resistance: a global threat, an international response
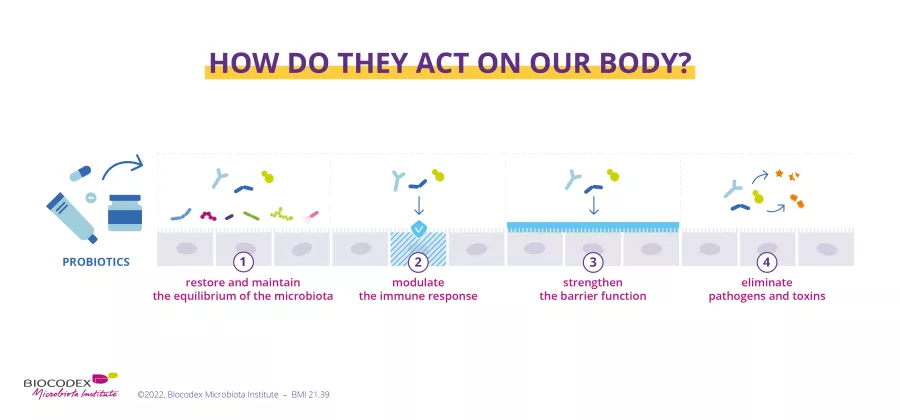
 Understanding the complex interplay between the microbiota and immunity is only just beginning
Understanding the complex interplay between the microbiota and immunity is only just beginning