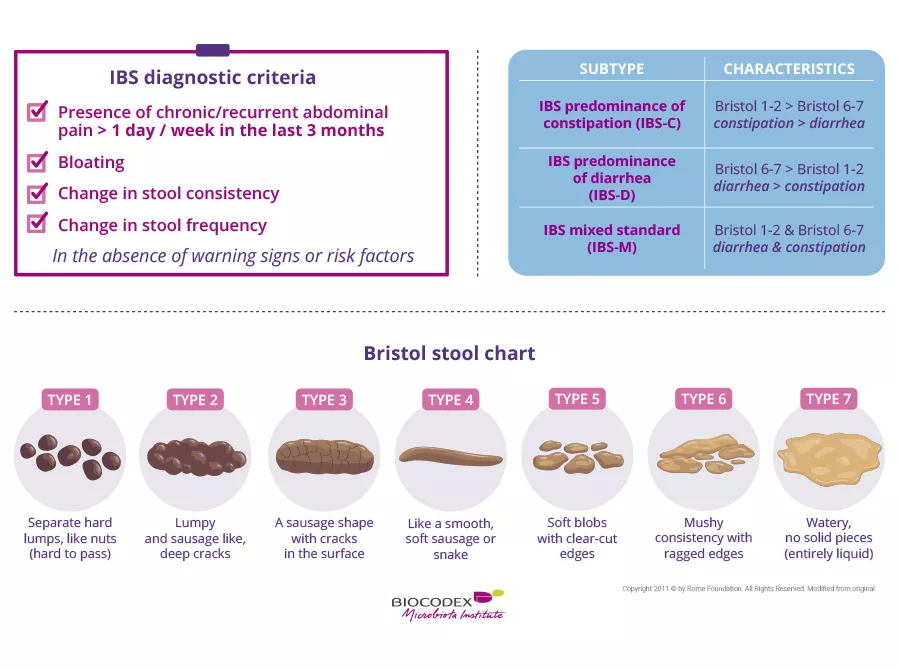
Plastic takeaway packaging releases microplastics and nanoplastics that may have a significant impact on the gut and oral microbiota.
Harmful effects on humans remain under-investigated
A study carried out in 2020 showed that plastic food containers made from polypropylene (PP), polystyrene (PS), polyethylene (PE), and polyethylene terephthalate (PET) all contain micro- and nanoplastics. Although the harmful effects of these particles on the microbiota have been demonstrated in animals (fish, shrimp, and mice), until now they have been under-investigated in humans.
Researchers analyzed and compared the gut and oral microbiota of 390 Chinese students aged between 18 and 30 who consumed takeaway food served in plastic packaging either frequently (at least three times a week), occasionally (once a week or less), or never.
The scientists also created three groups of mice which, for five weeks, were given either a solution of microplastics at 5 mg/ml (20 mice), a solution of nanoplastics at 5 mg/ml (20 mice), or a solution of the same nanoplastics but at 2 mg/ml (20 mice). A control group of 15 mice was also established.
Disruption of microbial balance
The results show that consumers of takeaway food in plastic containers more frequently suffer from gut disorders and coughs than those who never consume such food. They also presented a dysbiosis of the gut and oral microbiota with specific bacterial signatures. The gut microbiota of occasional consumers was strongly associated with the presence of Faecalibacterium, while that of heavy consumers was associated with Collinsella. For the oral microbiota, the bacteria most strongly associated with heavy consumers was Thiobacillus.
Significant impact even with smaller particles and at low doses
The study on mice showed that all the animals fed with plastic particles presented a gut dysbiosis when compared to the control group. Changes in the size (i.e. micro- vs. nanoplastics) or quantity (5 mg/ml vs. 2 mg/ml) of the particles ingested had no effect on their impact.
Moreover, the researchers believe that the recurrent cough in takeaway lovers may mean that micro- and nanoplastics are able to migrate from the gut to the lungs, where they accumulate in the airway microbiota, causing a dysbiosis that leads to the cough. Although yet to be confirmed, these results are one more reason to switch to a glass lunch-box.

 Could fibers modify the microbiota?
Could fibers modify the microbiota?
 Green Mediterranean diet: what links between cardiometabolic health and gut microbiota?
Green Mediterranean diet: what links between cardiometabolic health and gut microbiota?