Human milk nutrient fortifiers alter the developing gastrointestinal microbiota of very-low-birth-weight infants
Commented article - Children's section
By Pr. Emmanuel Mas
Gastroenterology and Nutrition Department, Children’s Hospital, Toulouse, France
Comment on the article by Asbury et al. (Cell Host Microbe) [1]
Nutrient fortifiers are added to human milk to support the development of verylow- birth-weight infants. At present, bovine-milk-based fortifiers (BMBFs) are predominantly administered, but there is an increasing interest in adopting humanmilk- based fortifiers (HMBFs). Although beneficial for growth, their effects on the gastrointestinal microbiota are unclear. This triple-blind, randomised clinical trial (NCT02137473) tested how nutrient-enriching human milk with HMBFs versus BMBFs affects the gastrointestinal microbiota of infants born <1,250 g during hospitalisation. These results highlight how nutrient fortifiers impact the microbiota of very-low-birth-weight infants during a critical developmental window.
WHAT DO WE ALREADY KNOW ABOUT THIS SUBJECT?
Breast milk (BM) is recognised as the best choice for feeding infants, especially those with a very-low-birth-weight (VLBW), i.e. <1,250 g. In intensive care units, when breastfeeding is impossible, it is recommended using pasteurised human breast milk (PHBM) donated from a breast-milk bank. BM or PHBM often requires enrichment to ensure optimal growth. Bovine milk- based fortifiers (BMBFs) have traditionally been used for enrichment; however, more recently, human-milk-based fortifiers (HMBFs) are also used. While it is well established that VLBW infants have abnormal gut microbiota, it is not known how to improve the composition of this gut microbiota with the nutrients used in VLBW infants.
Clinical studies are needed to determine the impact of these different enrichments on the gut microbiota of VLBW infants.
WHAT ARE THE MAIN INSIGHTS FROM THIS STUDY?
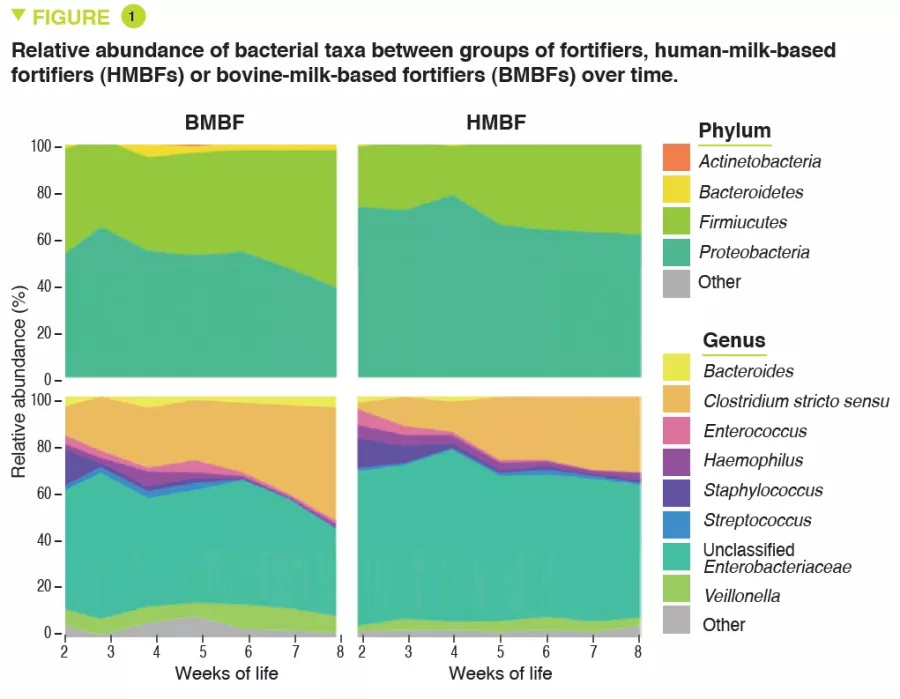
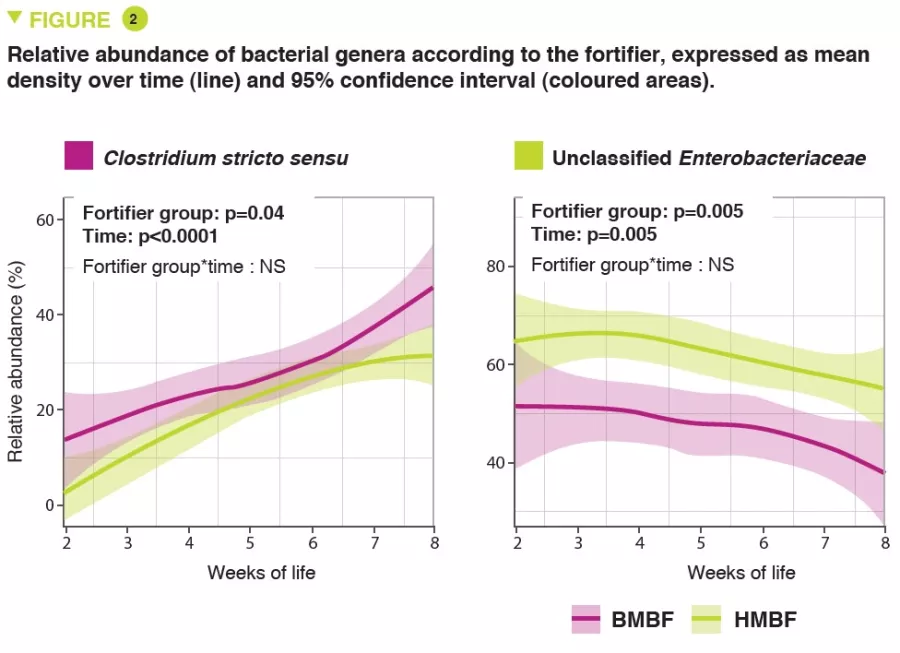
The OptiMom randomised controlled trial included 119 infants with a birth weight <1,250g (56 BMBFs and 63 HMBFs). The median term and birth weight were 880 g and 27.9 weeks, with no differences in any of the parameters between the two groups. HMBF infants had lower microbial diversity (Shannon index) (p <0.005). A predominance of Proteobacteria and Firmicutes was observed in both groups, with a higher relative abundance of Proteobacteria (p = 0.0003) including unclassified Enterobacteriaceae (p = 0.005) and a lower abundance of Firmicutes (p = 0.001) including Clostridium stricto sensu (p = 0.04) in HMBFs compared to BMBFs (Figure 1). Bacterial abundance increased steadily over time in the BMBF group but changed little in the HMBF group (p = 0.03). The relative abundance of Clostridium stricto sensu (p = 0.04) was higher in BMBF infants compared to HMBF infants, and unclassified Enterobacteriaceae were lower (p = 0.005)(Figure 2). After normalising the abundance of taxa, other differences emerged on a genus level with higher concentrations of unclassified Eubacteriaceae (p <0.0001), Streptococcus (p = 0.0002) and Staphylococcus (p = 0.002), and lower concentrations of Clostridium stricto sensu (p = 0.04) in HMBF infants compared to BMBF infants. These changes in bacterial abundance were associated with changes in microbial function. Finally, it was possible to predict the type of fortifier received based on microbial abundance in stools.
The authors were interested in the effects of milk volumes. In both groups, higher volumes of BM for three days were associated with higher alpha diversity but were unrelated to total bacterial density. With higher BM volumes, a higher relative and normalised abundance of Veillonella was observed in both groups, and Streptococcus in the BMBF group. A positive link between BM volumes and Staphylococcus concentrations was observed in the HMBF group, and with unclassified Eubacteriaceae in the BMBF group.
PHBM volumes were only associated with higher diversity in the BMBF group and bacterial density. Similarly, lower relative and standardised abundances of unclassified Eubacteriaceae, Streptococcus and higher abundance of Clostridium stricto sensu were reported in BMBF infants with higher PHBM volumes.
Higher volumes of BMBF were positively related to bacterial diversity and density in the BMBF group but not in the HMBF group. BMBF volumes were positively associated with relative and standardised abundances of Firmicutes and Clostridium stricto sensu, whereas HMBF volumes were positively associated with relative and standardised abundances of Clostridium stricto sensu and negatively associated with Staphylococcus.
Key point
- The use of human-milk based fortifiers or bovinemilk based fortifiers in the diet of very-low-birth-weight infants alters differently the bacterial composition of the gut microbiota during the first weeks of life
WHAT ARE THE CONSEQUENCES IN PRACTICE?
This study shows the importance of understanding the impact of the different nutrients used in the gut microbiota of VLBW infants to achieve a beneficial effect on their shortand long-term health.
Conclusion
This study showed that nutritional fortifiers alter the development of the gut microbiota in very-low-birthweight infants. It also showed that associations exist between amounts of enteral nutrition components in these children, BM, PHBM and bacterial communities.