Can low-calorie sweeteners such as aspartame, sucralose, or acesulfame be part of a diet for managing overweight or obesity? This topic is controversial.
Contradictory scientific data
While some studies have raised concerns that have led the WHO to advise against their use for weight loss and health improvement1, several long-term clinical trials suggest that these additives are at worst harmless and at best beneficial in these areas.
The same problem applies to microbiota: some studies suggest they have adverse effects on certain aspects of the gut microbiota linked to glycemic response, while others show the opposite, and still others show that it is mainly sugary drinks that impact microbiota and metabolites associated with diabetes risk.
A real-world study settles the matter
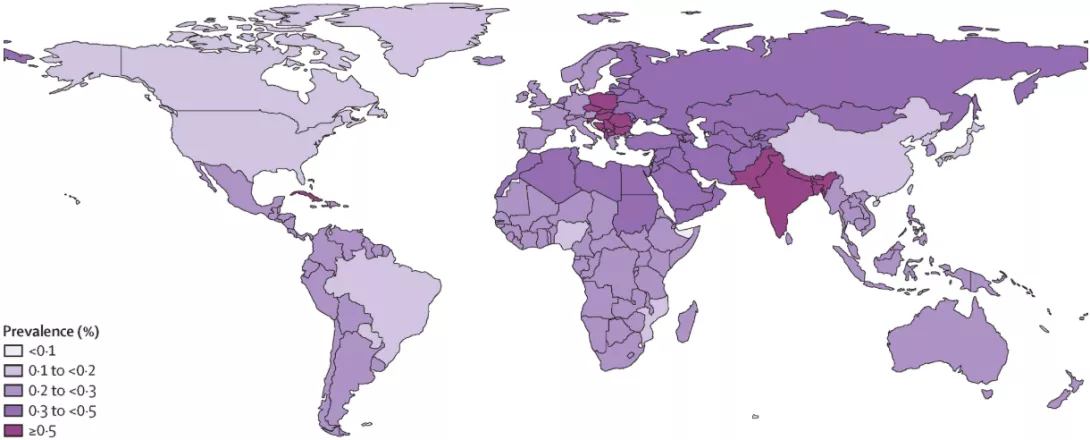
To try shed some light on the matter, a team of researchers recruited 341 overweight or obese individuals (average BMI of 31; 70% women, average age 47) living in Denmark, Greece, Spain, and the Netherlands. Their goal was to test the real effect of replacing sugar with sweeteners after weight loss.
The volunteers first followed a low-calorie diet for two months, then those who lost more than 5% of their body weight (277 people) adopted a healthy, balanced diet without quantity restrictions for the following 10 months.
Half of them consumed sweetened products instead of sugar-rich products (the “sweeteners” group); the other half received conventional sweetened products amounting to less than 10% of their total energy intake, in accordance with WHO recommendations (the “sugar” group).
The researchers analyzed changes in body weight and cardiometabolic markers in all participants, as well as the microbiota composition of a sample of 137 individuals from both groups.
Conclusion
This high-quality study (multicenter, long-term, real-world conditions, etc.) provides evidence that the prolonged use of sweeteners as part of a healthy, low-sugar diet can contribute to weight loss without adverse effects on cardiometabolic parameters or the gut microbiota.
Now you know!

 Microbiota and exposome: a dialog at the core of our health
Microbiota and exposome: a dialog at the core of our health
 Infant microbiota: it’s up to you, dads!
Infant microbiota: it’s up to you, dads!