Depression: when sodas ruin your gut flora and your mood
In women, high soda consumption is associated with more frequent and severe depressive disorders. An intestinal bacterium, Eggerthella, may partly explain this link.
The gut microbiota
In women, high soda consumption is associated with more frequent and severe depressive disorders. An intestinal bacterium, Eggerthella, may partly explain this link.
The gut microbiota
The vaginal microbiota is a dynamic reservoir of antibiotic resistance genes, whose presence is linked to increased microbial diversity and bacteria associated with vaginal dysbiosis, all of which are influenced by lifestyle.
 6 things you should know about antibiotics
6 things you should know about antibiotics
 Keys to a successful consultation by Alessandra Graziottin
Keys to a successful consultation by Alessandra Graziottin
Basil essential oil could soon be counted among the natural treatments for vaginal infections. According to a new study, the oil contains compounds capable of eradicating pathogens while respecting the balance of the vaginal microbiota.
Vaginal infections Vaginal infections Vaginal yeast infection
 Good eating habits to take care of your vaginal flora
Good eating habits to take care of your vaginal flora
 Vaginal microbiota and human papillomavirus (HPV): bacteria to the rescue
Vaginal microbiota and human papillomavirus (HPV): bacteria to the rescue
A single gut microbe may influence whether a patient responds to cancer immunotherapy. This study reveals how signals from the intestine can strengthen the body’s antitumour defences in ways we never expected.
 Gut microbiota and chemotherapy: adverse effects or better treatment efficacy?
Gut microbiota and chemotherapy: adverse effects or better treatment efficacy?
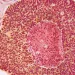 Cancer: microbiota involved in the abscopal effect
Cancer: microbiota involved in the abscopal effect
What is the exposome and how does it interact with the human microbiota? In this video series with Dr Véronique Mondain, discover how the exposome connects the microbiota, the body and the environment, and why it is essential for the future of healthcare.
Exposome: exposed microbiota puts health at risk Diet The gut microbiota The vaginal microbiota
Anti-inflammatory diets support a healthy oral and gut microbiota, leading to better cognitive performance... and perhaps helping prevent cognitive decline?
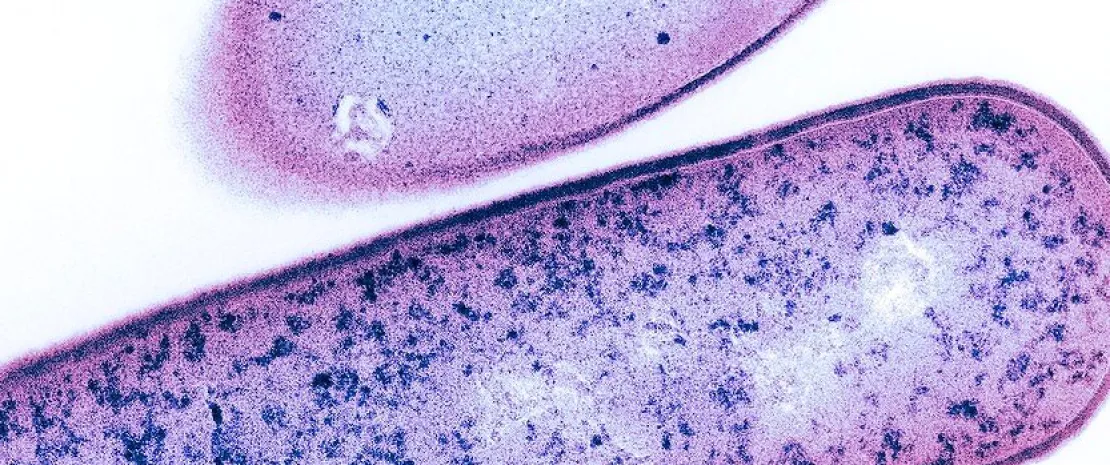
 Diet, microbiota, and the immune system acting together in metabolic syndrome
Diet, microbiota, and the immune system acting together in metabolic syndrome
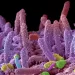 How oral microbiome metabolites drive gum disease & inflammation
How oral microbiome metabolites drive gum disease & inflammation
Did you think the Mediterranean diet was good only for your heart (and your taste buds)? Good news: it may also nurture your brain, through a rather unexpected path – your oral and gut microbiota!
The gut microbiota
 The surprising key to Alzheimer's may be in your gut, not your brain
The surprising key to Alzheimer's may be in your gut, not your brain
What if the microbes in a woman’s gut and vagina could predict her stress, sexual well-being, or recovery from cancer? A new study reveals the microbiome isn’t just a passenger, it may be an unseen driver of quality of life in endometrial disease.
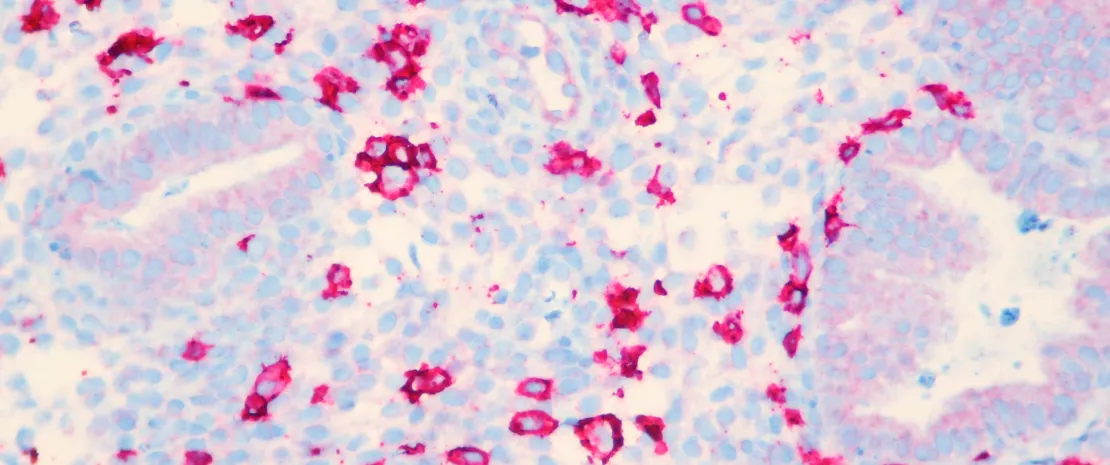
Before multiple sclerosis progresses, the gut may whisper its warning. New research reveals that specific microbes and their metabolites vanish years before neurodegeneration accelerates, offering clinicians a metabolic early-warning system.
Neurological disorders
 Multiple sclerosis and microbiota: does meat consumption play a role?
Multiple sclerosis and microbiota: does meat consumption play a role?
 Specific microbial species correlate with amyloid and tau pathology in Alzheimer’s disease
Specific microbial species correlate with amyloid and tau pathology in Alzheimer’s disease