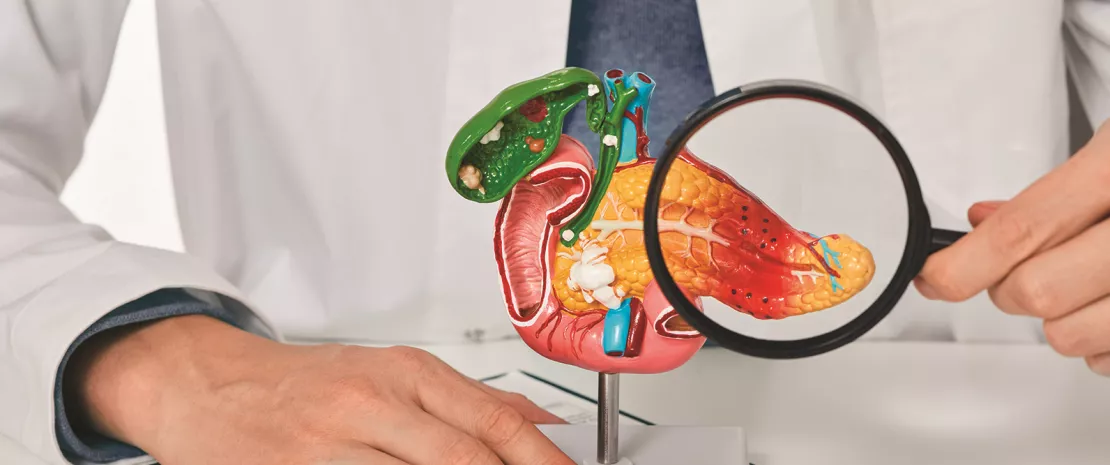
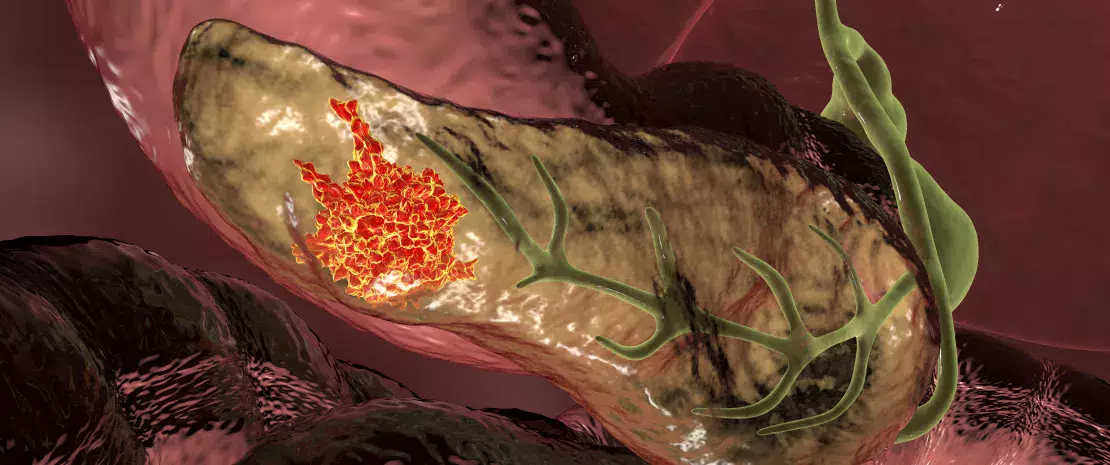
Pancreatic cancer remains one of the most deadly cancers, with a five-year survival rate of around 13%. Established risk factors (smoking, obesity, pancreatitis, and genetics) account for only 30% of cases.
Several epidemiological studies have shown that poor oral health, particularly the presence of periodontal disease and oral candidiasis, is associated with this cancer. Despite this, our knowledge about the actual involvement of oral microorganisms remains limited.
A large-scale prospective study using cohorts from the American Cancer Society Cancer Prevention Study-II and the Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial, has recently revealed that the oral microbiota, particularly bacteria and fungi, may play a significant role in the subsequent development of pancreatic cancer.
Periodontal bacteria: a strong link
Of the 122,000 participants who provided an oral sample, 445 developed pancreatic cancer during nearly nine years of follow-up. The researchers matched them with 445 healthy individuals to compare their oral microbiota.
The results published in JAMA Oncology 1 show that three major pathogenic bacteria known to be involved in periodontal disease (Porphyromonas gingivalis, Eubacterium nodatum, Parvimonas micra) significantly increase the risk.
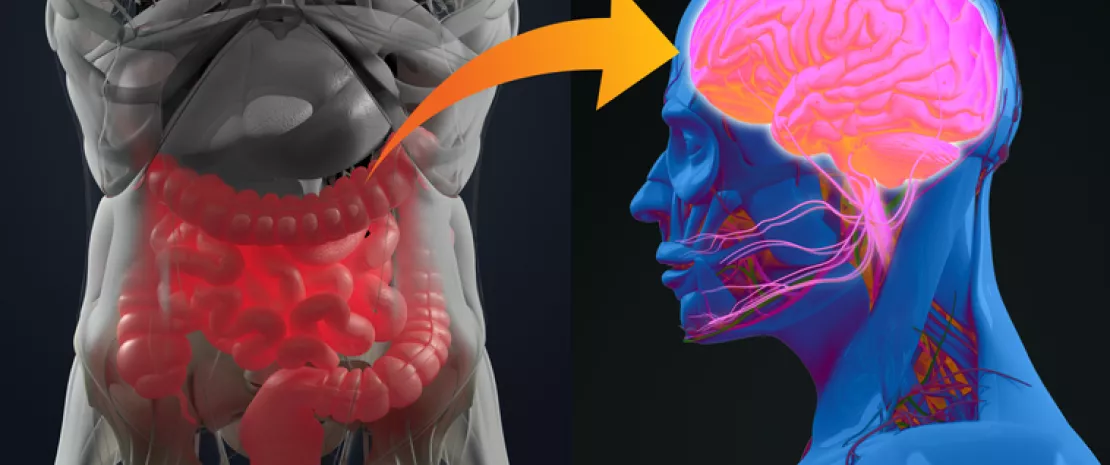
A comprehensive analysis of bacterial genomes revealed that a total of 21 species influence the risk of pancreatic cancer, some protective, others harmful. These taxa are thought to be associated with metabolic pathways that may promote the neoplastic transformation of pancreatic cells during their migration from the mouth to the pancreas.
Candida at the forefront
An analysis of the oral fungal microbiota—a parameter rarely explored in this context—also revealed that the genus Candida is associated with an increased risk of pancreatic cancer.
Candida tropicalis and Candida spp are associated with an increased risk, while C. albicans shows an inverse relationship. Within the Malassezia genus, only M. globosa was associated with cancer, and its effect was protective.
The researchers also report having confirmed the presence of Candida in biological samples of cancerous pancreatic tissue in cancer patients. This again supports the hypothesis of a migration of this fungus to the tumor and a direct role in carcinogenesis.
Towards early diagnosis and a better understanding of risk factors
In order to assess the impact of these microorganisms as a whole, the researchers calculated a Microbial Risk Score (MRS) for each participant, incorporating 27 bacterial and fungal species associated with cancer. They found that each increase of one standard deviation in the MRS more than triples the risk of pancreatic cancer (odds ratio [OR] 3.44).
According to the authors, the MRS score was reproducible between the two cohorts, suggesting the oral microbiota could one day be used as a biomarker in primary prevention to screen high-risk patients.
More generally, the data from this study reinforce the hypothesis that oral health is an important factor in the prevention of pancreatic cancer. Now we must try to better understand how oral fungal and bacterial communities promote cancerization processes.
To be continued...
 IBD and pregnancy: importance of vaginal microbiota
IBD and pregnancy: importance of vaginal microbiota
 ART and embryo transfer: is your vaginal flora one of the keys to success?
ART and embryo transfer: is your vaginal flora one of the keys to success?