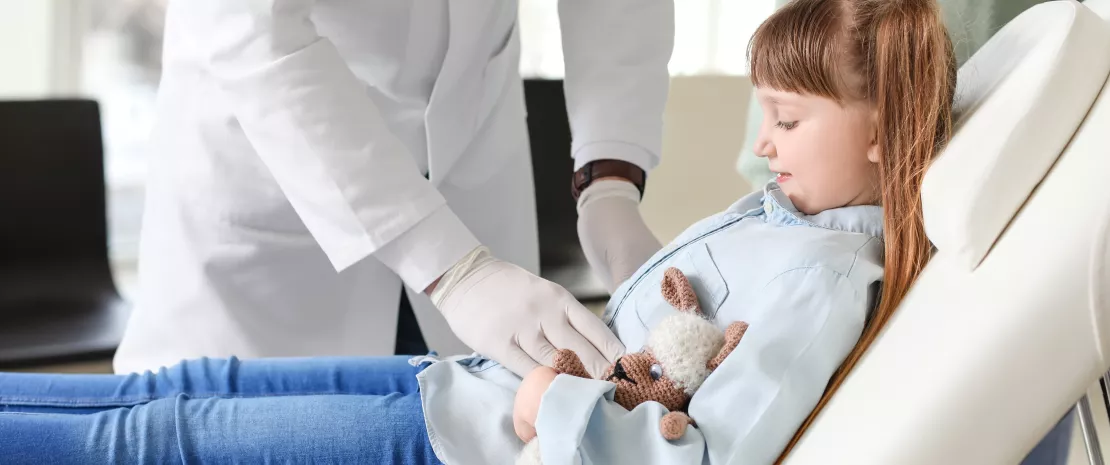
Antibiotics not only eradicate pathogenic microorganisms responsible for infection, but they can also destroy some bacteria that are beneficial to the gut microbiota. They always cause a more or less significant imbalance within this ecosystem. This ecosystem becomes less rich and less diversified, and is no longer able to properly carry out its protective functions2.
Antibiotics are not automatic anymore15 !
Between 10 and 30% of patients treated with antibiotics experience a change in their intestinal transit within 3-5 days of treatment, most often in the form of diarrhea16. Most of the time, this diarrhea is purely functional, caused by the antibiotic-induced dysbiosis. It usually presents without fever, it is short lived, not severe, and in most cases, it regresses once antibiotics are discontinued, or in the following weeks. However, dysbiosis starts in the first 24 hours of antibiotic treatment and lasts up to six weeks after its conclusion. New microorganisms, close to the initial strains but not necessarily identical, slowly re-colonize the intestines and create a new balance, although it often remains incomplete.
A new route for pathogens
But sometimes, the mucus layer, true line of defense of our intestines, has been so weakened that the body becomes more vulnerable to pathogens. In 10 to 20% of cases, diarrhea results from the colonization of the microbiota by Clostridium difficile. This bacterium is very widespread in hospitals and nursing homes, and elderly people are particularly vulnerable: in these facilities, up to 20% of residents (50% in cases of long-term stay) can host this bacterium in their intestines without showing any symptom (asymptomatic carriers). Although most cases of C. difficile diarrhea resolve once the antibiotics are discontinued, most severe forms could occur (pseudomembranous colitis and fulminant colitis, especially in people over 65 years old17). In the US, for instance, this infection is responsible for around 30,000 deaths/year18. The families of antibiotics that are often blamed are penicillins, some generations of cephalosporins, fluoroquinolones and clindamycins. To stop this vicious circle, stool transplant (transplant of healthy microbiota into the GI tract of a patient) could be a therapeutic alternative to antibiotics in order to repopulate the microbiota and restore the gut barrier.