Understanding the complex interplay between the microbiota and immunity is only just beginning
By Dr Bruce Vallance


By Dr Bruce Vallance

Your body is home to trillions of bacteria that together with viruses, fungi and other organisms, collectively make up the human microbiota.
These microbes play an important role in promoting our health, as well as controlling our susceptibility to disease, by influencing different aspects of our daily lives. For example, the metabolic activity of our gut microbiota determines whether certain medications like acetaminophen are toxic to our livers.1 Specific members of the microbiota can also change and evolve in response to new dietary sources of carbohydrates, allowing us to digest foods like sushi2 or produce important and protective chemicals such as short chain fatty acids (SCFA).3 Other microbes selectively shape our immune systems to become reactive, or tolerant to invading organisms, thereby controlling our risk of severe gastrointestinal (GI) infections.4
During the 1000 first days of life, the critical window of early childhood growth and development (period from conception to 2 years of age), any interference with microbiota establishment in the neonatal gut may potentially lead to negative health outcomes.5
1000 first days of life critical window of early childhood growth and development
Although scientists have established the importance of the microbiota in maintaining human health, our understanding of the complex interplay between the microbiota and immunity is only just beginning.
A new study reveals that faecal microbiota transplantation improves the symptoms of irritable bowel syndrome, as well as patient quality of life, even one year after treatment. However, it is essential that the donor is in good health.
The gut microbiota Functional gastrointestinal disorders Fecal transplant
Pain, cramps, bloating, diarrhoea, constipation: irritable bowel syndrome is a disease characterised by a range of abdominal symptoms that come and go throughout the patient's life. These symptoms can be exacerbated by stress, changes in emotional state and certain foods, and have a considerable impact on patients' quality of life. While there are no anatomical or structural problems with the bowels of people who suffer from this syndrome, the finger is often pointed at the gut microbiota.
The researchers involved in this study performed a clinical trial to test the efficacy of faecal microbiota transplantation by using stool samples from a single Caucasian man aged 36 years1 who ticked all the "super donor" boxes: in good health, normal BMI, undertaking regular exercise, born vaginally and breastfed. Even better, he was not taking any medication, had only received three antibiotic treatments during the course of his life and was regularly taking dietary supplements. In this clinical trial, faecal microbiota transplantation was found to be effective in patients with irritable bowel syndrome. However, these results were only observed three months after the transplantation, and several questions still need to be answered, particularly whether the clinical effects of the transplantation continue into the long term. In the current study, the researchers continued to monitor these patients for one year.
Most of the patients who responded to faecal microbiota transplantation after three months maintained their response after one year. Another encouraging result was also observed: their abdominal symptoms, fatigue and quality of life were clearly improved compared to three months after the transplantation. Even better, between 32 and 45% of the patients, depending on the group, experienced complete remission over the course of the one-year follow-up. Complete analysis of the patients' gut microbiota revealed changes in the gut bacteria profile and a significant reduction in the dysbiosis index.
In conclusion, faecal microbiota transplantation from a "super donor" can restore the gut microbiota and reduce the symptoms of patients suffering from irritable bowel disease.
An incredible medical breakthrough becomes the victim of its own success. Since they were first discovered in the early 20th century, antibiotics have saved millions of lives. However, the wide scale and sometimes inappropriate use of these drugs is making them less and less effective at treating infections. The result is that many bacteria have now become resistant! But their excessive and incorrect use cannot take the whole blame for antibiotic resistance.
One study1 has revealed that international travel is increasing the acquisition of antimicrobial resistance genes and could be helping the spread of antibiotic resistance. Head straight to the departure lounge for more information.

Every year since 2015, the World AMR Awareness Week (WAAW) has been raising awareness of the rise in (sidenote: Antimicrobial Resistance Antimicrobial Resistance occurs when bacteria, viruses, fungi, and parasites change over time and no longer respond to medicines. Antibiotics and other antimicrobial medicines stop working, and infections become harder and even impossible to treat. This resistance to microbes increases the risk of disease spread, severe illness, and death. Antibiotic resistance means the resistance of bacteria to antibiotics. Source: Antimicrobial resistance. October 26, 2020. ) . Linked to the incorrect and overuse of antibiotics, this is when a bacterium is able to resist the action of an antibiotic. In 2020, the WAAW broadened the scope of its message to include (sidenote: Antimicrobials Medicines – including antibiotics, antivirals, antifungals and antiparasitics – used to prevent and treat infections in humans, animals and plants. WHO Antimicrobial Resistance; Nov 2023 ) - antivirals, antifungals and antiparasitics, etc., that is medicines that are essential for fighting (sidenote: Pathogens A pathogen is a microorganism that causes, or may cause, disease. Pirofski LA, Casadevall A. Q and A: What is a pathogen? A question that begs the point. BMC Biol. 2012 Jan 31;10:6. ) (sidenote: Microorganisms Living organisms too small to see with the naked eye. This includes bacteria, viruses, fungi, archaea, protozoa, etc., collectively known as ’microbes’. Source: What is microbiology? Microbiology Society. ) . By reducing our options for treating infections, antibiotic resistance has become a threat to everyone’s health2. Several studies are therefore underway to understand how this phenomenon is spreading so we can better control it, or even stop it altogether.
Antibiotics save life! Did you know that they also have an impact on your microbiota? Did you know that the misuse and overuse of antibiotics can lead to antibiotic resistance? Have you heard about the World AMR Awareness Week
(WAAW)? All the answers in this dedicated page:
We now know that antibiotic resistance is mainly due to the overuse of antibiotics in human medicine, as well as in livestock and agriculture2. However, a recent study has revealed an unexpected cause of this spread: holidays and business trips to far-flung destinations! In fact, international travel encourages the spread of (sidenote: Gene A gene is the basic physical and functional unit of heredity. Genes are made up of DNA. Source: What is a gene? MedlinePlus.gov. ) in the gut. Researchers studied 190 Dutch travelers, divided into four subgroups based on destination. They were all destined for areas badly affected by antibiotic resistance, namely South East Asia, South Asia, North Africa, and East Africa The aim was to determine whether international travel to these regions could encourage the spread of resistance to areas less badly affected. In order to measure the abundance of these genes in the gut, a stool sample was taken from each participant before and after travel.

Using cutting edge technology (
(sidenote:
Metagenomics
A method of studying the genetic material in samples taken directly from complex natural environments (intestines, oceans, soil, air, etc.), as opposed to samples grown in a laboratory. It produces a description of the genes contained in the sample, as well as an insight into the functional potential of the microbial community.
Source: Riesenfeld CS, Schloss PD, Handelsman J. Metagenomics: genomic analysis of microbial communities. Annu Rev Genet. 2004;38:525-52.
)
), the team observed an increase in the number of antibiotic resistance genes between departure and return, especially in travelers coming back from South East Asia. A total of around fifty antibiotic resistance genes was detected following travel. These included genes with well-known and long-standing resistance to antibiotics (such as β-lactams, tetracyclines and fluoroquinolones) as well as new, never-before seen genes.
The results of this study are clear: international travelers, colonized by resistance genes whilst on their travels, could unwittingly be bringing back antibiotic-resistant bacteria along with the rest of their luggage. Faced with the risk of this resistance spreading, the authors sound the alarm and highlight the importance of taking rapid action in countries particularly affected by antibiotic resistance. This resonates with the WHO's campaign.
Each year, since 2015, the WHO organizes the World AMR Awareness Week (WAAW), which aims to increase awareness of global antimicrobial resistance.
Antimicrobial resistance occurs when bacteria, viruses, parasites and fungi change over time and no longer respond to medicines. As a result of drug resistance, antibiotics and other antimicrobial medicines become ineffective and infections become increasingly difficult or impossible to treat, increasing the risk of disease spread, severe illness and death.
Held on 18-24 November, this campaign encourages the general public, healthcare professionals and decision-makers to use antibiotics, antivirals, antifungals and antiparasitics carefully, to prevent the further emergence of antimicrobial resistance.
 6 essential things you should know about antibiotics
6 essential things you should know about antibiotics
 Antimicrobial resistance: a universal threat, an international response
Antimicrobial resistance: a universal threat, an international response
Dysbiosis refers to a breakdown in the delicate balance between the billions of microorganisms that make up our human microbiota and in their relationship with our body. There are a range of factors that lead to dysbiosis, including genetics, an unbalanced diet and antibiotics, with most cases involving several factors. Today, scientific research has demonstrated that dysbiosis of the most extensively studied microbiota—the gut microbiota—as well as other microbiotas such as the vaginal, skin and lung microbiotas, is associated with various diseases, from irritable bowel syndrome to metabolic disorders like obesity, or even chronic sinusitis and eczema. But how does the microbiota become imbalanced? What impacts does dysbiosis have on our health? How can we rebalance the microbiota?
Read on to find out!

First, let's look at the word itself: "dysbiosis". The etymology of this scientific term is actually very straightforward! In Greek, the word bios means "living" and the prefix dys- means "bad".
"Dysbiosis" can be defined as a change in the composition and functioning of the microbiota. This change is the result of a combination of environmental factors and factors specific to each person1.
Our gut microbiota is the human body's main microbiota.2 It contains at least 1000 different species3 of microorganisms, including bacteria, fungi and viruses. The Firmicutes group (which includes the well-known "good bacteria", lactobacilli) and the Bacteroidetes group together account for 70–90% of the bacterial community in our gut.2,4 Our microbiota also contains Actinobacteria, which include bifidobacteria, known for their beneficial effects. Other microorganisms in our microbiota can make us ill. These are referred to as " (sidenote: Pathogen A pathogen is a microorganism that causes, or may cause, disease. Pirofski LA, Casadevall A. Q and A: What is a pathogen? A question that begs the point. BMC Biol. 2012 Jan 31;10:6. ) ", but they are in the minority.2 Dysbiosis involves one or more of the following phenomena:
As a result, our microbiota is weakened and the "bad" bacteria take over from the "good" bacteria.2 It becomes harder for the microbiota to protect the body from attack and to carry out its essential roles relating to our health and well-being.1,6
1000 It contains at least 1000 different species of microorganisms.
While dysbiosis itself is not considered a disease, it has been linked to various health problems and may contribute to the development or exacerbation of certain medical conditions.
However, "dysbiosis" is not a universal term that can be applied to anyone in any circumstance! 1 In fact, the composition of our microbiota is particular to each person, and is influenced by our genes and by the microorganisms that colonised our body during our first years of life ("microorganisms" are defined as "living organisms that are too small to be visible with the naked eye. This includes bacteria, viruses, fungi, archaea, protozoa and so on, collectively known as ' (sidenote: https://microbiologysociety.org/why-microbiology-matters/what-is-microbiology.html ) '). The microbiota varies so considerably between individuals that it could well be as unique to each of us as our fingerprints.7 What's more, it can change depending on our age, health, stress levels and diet, as well as where we live and any medicines we are taking.8 This means that each of us can experience our own specific dysbiosis when our microbiota becomes imbalanced and no longer functions correctly within our body.1
The prefix dys- in "dysbiosis" is the opposite of eu- ("good") or sym- ("with"). So we talk about "eubiosis" or "symbiosis" when our microbiota is in good health: when it is interacting harmoniously with our body and its microbial community is in balance.1
The relationship between our body and the billions of microorganisms that make up our microbiota is mutually beneficial.9 Each has its role to play: the body provides "bed and board" for the microorganisms, while the latter contribute to numerous important functions within the body, including digestion, nutrient assimilation, keeping the intestinal walls impermeable and fighting unwanted germs.2,8,10 Teamwork in action!
The various microorganisms that populate the microbiota community — including potentially pathogenic ones — are present in sufficient numbers and proportions to cohabit peacefully and fulfil their beneficial functions within the body. However, the delicate balance between the microbial ecosystems within our body can be upset, at which point eubiosis turns to dysbiosis.8
As its definition suggests, dysbiosis occurs under the influence of numerous very different and often interrelated factors.5 Among these are the following:
What can cause a dysbiosis ?
Antibiotics were one of the most important therapeutic breakthroughs of the twentieth century. Since penicillin was discovered in 1928, they have saved millions of lives.13 However, by destroying "good" bacteria alongside harmful ones, they unbalance the microbiota. In the short term, dysbiosis caused by antibiotics can result in diarrhoea14 or vaginal thrush.15 Gut dysbiosis caused by antibiotics is also suspected to have a long-term impact — particularly when antibiotics are taken during childhood — increasing the risk of various chronic diseases such as obesity and allergies.16
During infections such as viral gastroenteritis or food poisoning caused by salmonella, harmful and aggressive microbes invade the human microbiota. They come not from the microbiota itself but from outside, transmitted on hands or contaminated food, for example. These infections provoke a strong response from our immune system, with inflammation of the gut and diarrhoea. All this results in a sudden disruption to the balance of our intestinal flora. What's more, the microbes that cause these infections can also stimulate the growth of other potentially pathogenic bacteria already present in the microbiota. Infections therefore lead to dysbiosis, which all harmful bacteria take advantage of!1,12,17,18
What we eat has an impact on our microbiota throughout our lives. Any sudden changes in diet, in terms of quantity or content, can trigger dysbiosis. But it doesn't need to be sudden: while normal variations in our meals from day to day cause only temporary changes to the microbiota, the type of food we eat can cause lasting changes to the gastrointestinal ecosystem5 and ultimately become a factor in dysbiosis. Studies suggest that "Western" diets rich in fat, sugar and protein make us more susceptible to imbalances of the gut microbiota, while varied diets rich in fruit and vegetables may protect the microbiota from dysbiosis.1,19
They have saved millions of lives but their excessive and inappropriate use has now raised serious concerns for health, notably with antibiotic resistance and microbiota dysbiosis. ach year, the WHO organizes the World AMR Awareness Week (WAAW) to increase awareness of antimicrobial resistance. Let’s take a look at this dedicated page:
Each year, since 2015, the WHO organizes the World AMR Awareness Week (WAAW), which aims to increase awareness of global antimicrobial resistance. Antimicrobial resistance occurs when bacteria, viruses, parasites and fungi change over time and no longer respond to medicines. As a result of drug resistance, antibiotics and other antimicrobial medicines become ineffective and infections become increasingly difficult or impossible to treat, increasing the risk of disease spread, severe illness and death. Held on 18-24 November, this campaign encourages the general public, healthcare professionals and decision-makers to use antibiotics, antivirals, antifungals and antiparasitics carefully, to prevent the further emergence of antimicrobial resistance.
Dysbiosis: a cause or a consequence of illness?
Numerous studies comparing the microbiotas of ill and healthy people have shown that dysbiosis is associated with a range of chronic conditions, including intestinal diseases such as irritable bowel syndrome and Crohn's disease, as well as obesity, allergies, asthma and certain cancers1. But is it the dysbiosis that causes the disease or the disease that causes the dysbiosis? According to scientists, the answer is still unclear, but numerous studies are currently under way to try and work it out.
In 2019, researchers launched Homo symbiosus, an extensive research project that aims to get to the bottom of this issue and give us a better understanding of why and how so many chronic diseases are associated with gut dysbiosis. The researchers' hypothesis is that "the phenomena of gut dysbiosis, microbial growth, inflammation and weakening of the intestinal wall" are mutually sustaining10.
Dysbiosis of the gut microbiota is associated with a variety of diseases, including gastrointestinal, metabolic22, allergic23 and even mental24 conditions. But the human body is also home to specific microbial ecosystems located in the skin25, urinary tract26, vagina27, mouth28 and lungs29, all of which can also become unbalanced and be associated with specific diseases.
Normally, after an episode of dysbiosis, the microbiota is able to naturally restore its initial balance (although it will never have exactly the same composition as before): it is said to be "resilient" 30. But sometimes, this "re-biosis", or return to microbial balance, can take time: even in a healthy adult, for example, it can take six months after taking antibiotics 31. Eventually, dysbiosis can lead to a prolonged state of imbalance that is self-sustaining over the long term, with the microbiota never fully returning to normal, which can be harmful to health 1 .
What should we do when dysbiosis strikes? There are several potential solutions to help us rebalance the microbiota and improve our health.
Probiotics are: "live microorganisms which when administered in adequate amounts confer a health benefit on the host".32,33 Here you will find a page dedicated to probiotics: how they work, how they are made and how to choose the right ones... Explore our page on probiotics.
Primarily derived from dietary fibre (fructo-oligosaccharides, galacto-oligosaccharides, inulin, etc.), prebiotics are indigestible nutrients or substrates that are used by the microorganisms in the microbiota and have positive effects on health.34,35 See here for more information on how they impact on the microbiota. Special products combining probiotics and prebiotics are called symbiotics.36,37
What we eat, as well as the quality and diversity of our food, affects the balance of our gut microbiota38,39, but can also impact its composition and, as a result, trigger the onset of certain diseases.22 Ask your GP and/or dietician for advice on which foods have beneficial or harmful effects, to keep your gut in the best possible shape 40 and stay healthy !
Just like other organs, the microbiota can be transplanted from one individual to another, to try and restore the balance of the recipient's microbial ecosystem.41,42 At present, this approach is well documented for the gut microbiota. It is known in this context as faecal microbiota transplantation (FMT), but is only authorised for the treatment of recurrent Clostridioides difficile infections.41 Intensive research is under way for other intestinal disorders.41 For the vagina, vaginal microbiota transplantation (VMT) is currently being trialled and could be a promising treatment for recurrent or refractory bacterial vaginosis 43. Studies into skin microbiota transplantation are still rare, but the initial results are promising.44,45

"Informative" -Peggy Rhinelander (From My health, my microbiota)
"Absolutely fascinating how dysbiosis reveals the hidden connections between our health and the microbiota!" -Aware Health Rewards App (From My health, my microbiota)

Since the discovery of penicillin in 1928, the widespread use of antibiotics has saved millions of lives. The main weapon in the fight against bacterial infections, antibiotics, alongside vaccinations, have added nearly twenty years to life expectancy.1
Gut, vagina, lungs, skin... many parts of the body play host to microorganisms (bacteria, fungi, viruses). Such microbial communities are known as microbiota.2 Antibiotics eradicate pathogenic germs responsible for infection but can also destroy certain beneficial bacteria in our microbiota, leading to imbalances ( (sidenote: Dysbiosis Generally defined as an alteration in the composition and function of the microbiota caused by a combination of environmental and individual-specific factors. Levy M, Kolodziejczyk AA, Thaiss CA, et al. Dysbiosis and the immune system. Nat Rev Immunol. 2017;17(4):219-232. ) 3) of varying degrees within these ecosystems. This concerns all of the body’s microbiota, including: the gut, skin,4 lung,5 ENT,6 urinary,7 and vaginal microbiota.8
By inducing a dysbiosis, antibiotics can have harmful effects on health. The main short-term complication is the alteration of bowel movements experienced by some patients. This most often results in diarrhea, with the gut microbiota less able to perform its protective functions. Antibiotic-associated diarrhea is usually mild to moderate9 in intensity, but its incidence varies according to age, type of antibiotic, context, etc. It may affect up to 35%9,10,11 of patients and 80% of children.9 In 10%-20% of cases, the diarrhea results from an infection by Clostridioides difficile (C. difficile),11 a bacterium that colonizes the gut microbiota and becomes pathogenic due to certain factors (e.g. antibiotic use). The clinical consequences vary, ranging from moderate diarrhea to much more serious symptoms, or even death.11
35% It may affect up to 35% of patients
80% and 80% of children
Diarrhea is not the only symptom of antibiotic-associated dysbiosis. When it occurs early in life, the condition is thought to be responsible for longer-term effects. The perinatal period, characterized by the development of the gut microbiota and the maturation of the immune system, is a particularly sensitive period.12 Antibiotic-associated dysbiosis during this phase seems to be a risk factor in the development of certain chronic diseases (obesity, diabetes mellitus, asthma, inflammatory bowel disease).13
Antibiotic resistance happens when an antibiotic treatment is no longer effective against a bacterial infection.1 The cause? Antibiotics are only effective against bacteria and have no effect on viruses (e.g. the flu).14 The inappropriate (e.g. with viral infections) or excessive use of antibiotics – in humans or animals – accelerates antibiotic resistance. Antibiotic resistance leads to longer hospitalizations, higher health care costs and more deaths. For this reason, the issue has become a major public health concern worldwide.1
Each year, from November 18 to 24, the WHO organizes World AMR Awareness Week, which aims to increase awareness of global (sidenote: Antimicrobial Class of drugs that includes antibiotics (active against bacteria), antiviral agents (active against virus), antiparasitic agents (active against parasites), and antifungal agents (active against fungi). WHO Antimicrobial Resistance; Oct 2020 ) resistance and to encourage best practices among the general public, health workers and policymakers to avoid the further emergence and spread of drug-resistant infections. As an expert on microbiota, the Biocodex Microbiota Institute takes part in this initiative.
Each year, since 2015, the WHO organizes the World AMR Awareness Week (WAAW), which aims to increase awareness of global antimicrobial resistance.
Held on 18-24 November, this campaign encourages the general public, healthcare professionals and decision-makers to use antimicrobials carefully, to prevent the further emergence of antimicrobial resistance.
1. WHO Antimicrobial Resistance; Oct 2020; https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
9. McFarland LV, Ozen M, Dinleyici EC et al. Comparison of pediatric and adult antibiotic-associated diarrhea and Clostridium difficile infections. World J Gastroenterol. 2016;22(11):3078-3104.
10. Bartlett JG. Clinical practice. Antibiotic-associated diarrhea. N Engl J Med 2002;346:334-9.
11. Theriot CM, Young VB. Interactions Between the Gastrointestinal Microbiome and Clostridium difficile. Annu Rev Microbiol. 2015;69:445-461.
14. Centers for Disease Control and Prevention; Patient Education and Promotional Resources https://www.cdc.gov/antibiotic-use/community/pdfs/aaw/au_improving-antibiotics-infographic_8_5x11_508.pdf
 The Janus face of Antibiotics: Life Savers and Microbiota Disruptors
The Janus face of Antibiotics: Life Savers and Microbiota Disruptors
 Antibiotic resistance is a web of several problems
Antibiotic resistance is a web of several problems
On the positive side, they are the mainstay in our therapeutic arsenal, saving millions of lives every year. On the flip side, they disrupt our microbiota and can have serious consequences on our health. Review of 6 key tips to use them wisely.

Since the discovery of penicillin in 1928, the widespread use of antibiotics has saved millions of lives. The main weapon in the fight against bacterial infections, antibiotics, alongside vaccinations, have added nearly twenty years to life expectancy.1

Gut, vagina, lungs, skin... many parts of the body play host to (sidenote: Microorganisms Living organisms that are too small to be seen with the naked eye. They include bacteria, viruses, fungi, archaea and protozoa, and are commonly referred to as “microbes”. What is microbiology? Microbiology Society. ) (bacteria, fungi, viruses). Such microbial communities are known as microbiota.2 Antibiotics eradicate pathogenic germs responsible for infection but can also destroy certain beneficial bacteria in our microbiota, leading to imbalances ( (sidenote: Dysbiosis Generally defined as an alteration in the composition and function of the microbiota caused by a combination of environmental and individual-specific factors. Levy M, Kolodziejczyk AA, Thaiss CA, et al. Dysbiosis and the immune system. Nat Rev Immunol. 2017;17(4):219-232. ) )3 of varying degrees within these ecosystems.
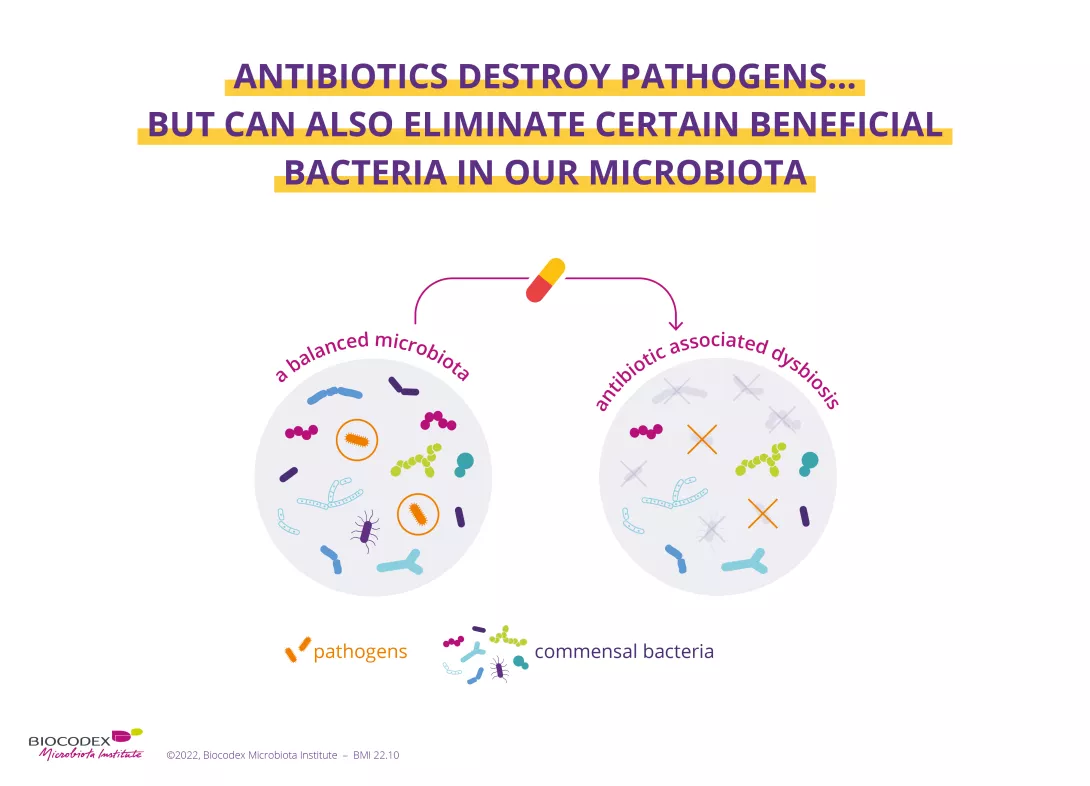
This concerns all of the body’s microbiota, including:
By inducing a dysbiosis, antibiotics can have harmful effects on health. The main short-term complication is the alteration of bowel movements experienced by some patients. This most often results in diarrhea, with the gut microbiota less able to perform its protective functions. Antibiotic-associated diarrhea is usually mild to moderate9 in intensity, but its incidence varies according to age, type of antibiotic, context, etc. It may affect up to 35%9,10,11 of patients and 80% of children.9 In 10%-20% of cases, the diarrhea results from an infection by Clostridioides difficile (C. difficile),11 a bacterium that colonizes the gut microbiota and becomes pathogenic due to certain factors (e.g. antibiotic use). The clinical consequences vary, ranging from moderate diarrhea to much more serious symptoms, or even death.11
35% Antibiotic-associated diarrhea may affect up to 35% of patients
80% and up to 80% if patients are children
Diarrhea is not the only symptom of antibiotic-associated dysbiosis. When it occurs early in life, the condition is thought to be responsible for longer-term effects. The perinatal period, characterized by the development of the gut microbiota and the maturation of the immune system, is a particularly sensitive period.12 Antibiotic-associated dysbiosis during this phase seems to be a risk factor in the development of certain chronic diseases (obesity, diabetes mellitus, asthma, inflammatory bowel disease).13
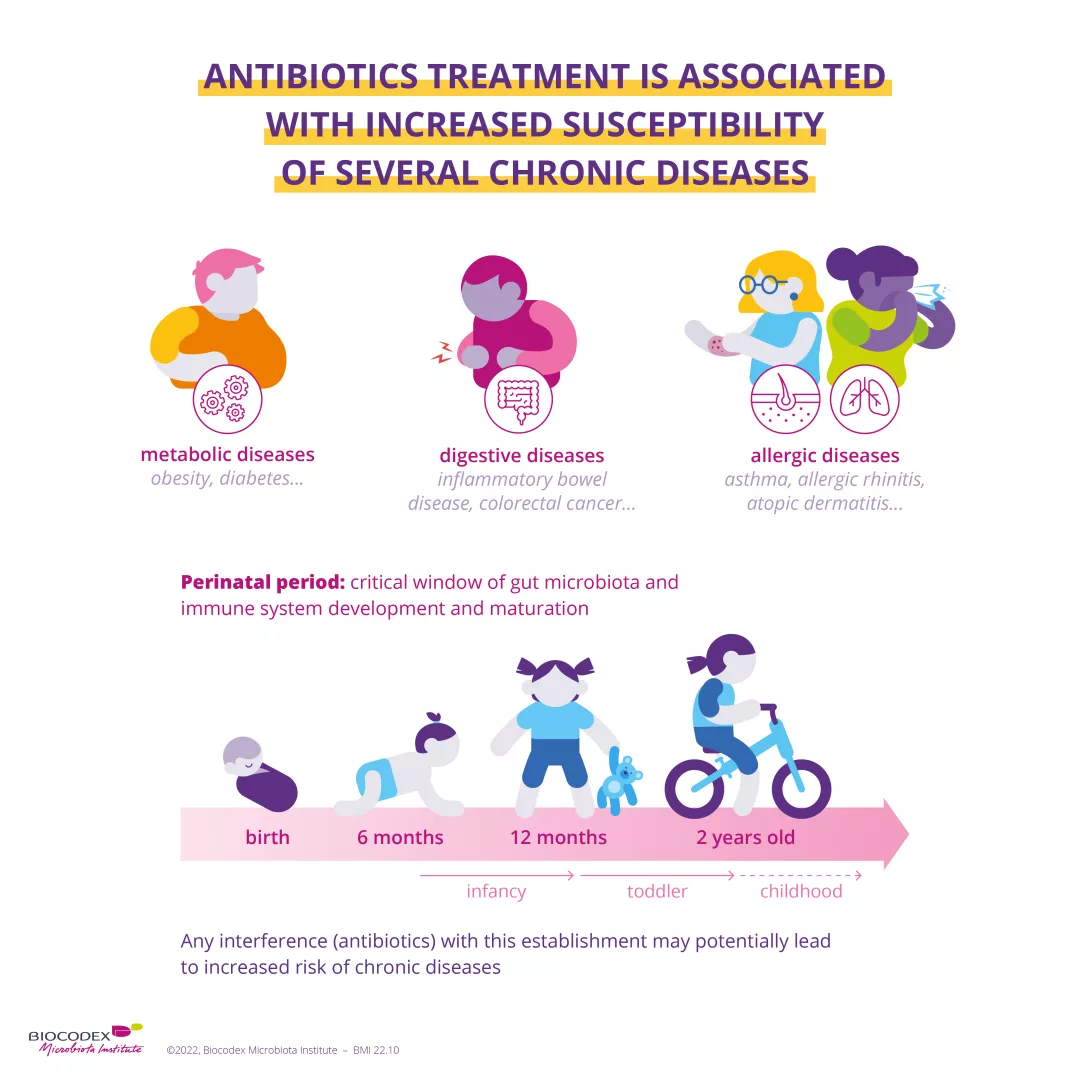
Antibiotic resistance happens when an antibiotic treatment is no longer effective against a bacterial infection.1 The cause? Antibiotics are only effective against bacteria and have no effect on viruses (e.g. the flu).14 The inappropriate (e.g. with viral infections) or excessive use of antibiotics – in humans or animals – accelerates antibiotic resistance. Antibiotic resistance leads to longer hospitalizations, higher health care costs and more deaths. For this reason, the issue has become a major public health concern worldwide.1

Each year, from November 18 to 24, the WHO organizes World AMR Awareness Week, which aims to increase awareness of global (sidenote: Antimicrobial Class of drugs that includes antibiotics (active against bacteria), antiviral agents (active against virus), antiparasitic agents (active against parasites), and antifungal agents (active against fungi). WHO Antimicrobial Resistance; Oct 2020 ) resistance and to encourage best practices among the general public, health workers and policymakers to avoid the further emergence and spread of drug-resistant infections. As an expert on microbiota, the Biocodex Microbiota Institute takes part in this initiative.
If you are interested in the effects of antibiotics on your health and your microbiota, or if you want to know more about the World AMR Awareness Week (WAAW), we recommend that you go to this other dedicated page:
Each year, since 2015, the WHO organizes the World AMR Awareness Week (WAAW), which aims to increase awareness of global antimicrobial resistance.
Antimicrobial resistance occurs when bacteria, viruses, parasites and fungi change over time and no longer respond to medicines. As a result of drug resistance, antibiotics and other antimicrobial medicines become ineffective and infections become increasingly difficult or impossible to treat, increasing the risk of disease spread, severe illness and death.
Held on 18-24 November, this campaign encourages the general public, healthcare professionals and decision-makers to use antibiotics, antivirals, antifungals and antiparasitics carefully, to prevent the further emergence of antimicrobial resistance.
1. WHO Antimicrobial Resistance; Oct 2020; https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance
9. McFarland LV, Ozen M, Dinleyici EC et al. Comparison of pediatric and adult antibiotic-associated diarrhea and Clostridium difficile infections. World J Gastroenterol. 2016;22(11):3078-3104.
10. Bartlett JG. Clinical practice. Antibiotic-associated diarrhea. N Engl J Med 2002;346:334-9.
11. Theriot CM, Young VB. Interactions Between the Gastrointestinal Microbiome and Clostridium difficile. Annu Rev Microbiol. 2015;69:445-461.
14. Centers for Disease Control and Prevention; Patient Education and Promotional Resources https://www.cdc.gov/antibiotic-use/community/pdfs/aaw/au_improving-antibiotics-infographic_8_5x11_508.pdf
 Antimicrobial resistance: a universal threat, an international response
Antimicrobial resistance: a universal threat, an international response
It could ultimately undermine a century of medical progress1. The ticking health time bomb of antimicrobial resistance is in sights of the WHO, which has organised the annual World AMR Awareness Week (18–24 November) since 2015. The Microbiota Institute plays an active role in this initiative, producing and sharing exclusive content throughout November on the impact of antimicrobials on the gut microbiota. We take a quick look at what's happening.

On the one hand, they are an extraordinary scientific discovery that saves millions of lives. On the other, their excessive and sometimes inappropriate use can lead to the emergence of multiple forms of resistance in microorganisms (including bacteria, viruses, parasites and fungi). As a result, although they were designed to heal, antimicrobials are becoming less and less effective and, ultimately, if no action is taken, there is a risk that they will no longer be able to protect us from infections.
10 million Antimicrobial resistance would become responsible for almost 10 million deaths worldwide by 2050
Antimicrobial resistance would become responsible for almost 700,000 annual deaths worldwide2. If nothing changes, infectious diseases could become one of the leading causes of mortality worldwide by 2050, causing up to 10 million deaths2.
The WHO is spearheading the international response to this scourge. Since 2015, it has organised World AMR Awareness Week, held on 18–24 November, which aims to increase awareness of this global phenomenon and encourage the general public, healthcare professionals and decision-makers to use antibiotics carefully, to prevent the further emergence of antimicrobial resistance.
As a major centre of expertise on the microbiota, the Microbiota Institute has been an active partner for the event since 2020. Throughout November, the Institute will be sharing articles and news, as well as expert videos and downloads on key topics, to enhance your knowledge and help you understand the mid- and long-term effects of antibiotics on the human microbiota. To cite just one example, despite their well-known efficacy against bacteria (and lack of efficacy against viral infection3), they often lead to dysbiosis. This is associated with several well-known problems, such as antibiotic-associated diarrhea.
Cornerstone of the modern therapeutic arsenal, antibiotics saved millions of lives. On the other hand, their excessive and sometimes inappropriate use can lead to the emergence of multiple forms of resistance in microorganisms. Each year, the World Health Organization (WHO) organizes the World AMR Awareness Week (WAAW) to increase awareness of this public health issue. Read the dedicated page:
But there's more! Taking antibiotics is also suspected to increase the risk of multiple chronic diseases4 (allergies, asthma, obesity, chronic inflammatory bowel disease, etc.), particularly if they are prescribed in early childhood. So can we do anything about it? Yes! By encouraging good prescription practices to ensure that antibiotics are used properly! But also by educating patients on the risks of dysbiosis associated with excessive and inappropriate use of antibiotics. We are all responsible and we all have a role to play in reducing antimicrobial resistance!
His team pioneered an ambitious study on the resistome of 700 children that will facilitate a breakthrough in the understanding of the evolution and dissemination of antimicrobial resistance in the early life human gut.
2 Tackling drug-resistant infections globally: final report and recommendations; May 2016.
3 Improving Antibiotic Use. Material Developed by CDC Using CDC materials does not imply endorsement or recommendation by CDC, ATSDR, HHS or the United States Government
 Antimicrobial resistance genes “stowaway” in gut microbiota during international travel
Antimicrobial resistance genes “stowaway” in gut microbiota during international travel
 Antibiotics: Dr Jekyll and Mr Hyde
Antibiotics: Dr Jekyll and Mr Hyde
It could ultimately undermine a century of medical progress1. The ticking health time bomb of antimicrobial resistance is in sights of the WHO, which has organised the annual World AMR Awareness Week (18–24 November) since 2015. The Microbiota Institute plays an active role in this initiative, producing and sharing exclusive content throughout November on the impact of antimicrobials on the gut microbiota. We take a quick look at what's happening.

On the one hand, they are an extraordinary scientific discovery that saves millions of lives. On the other, their excessive and sometimes inappropriate use can lead to the emergence of multiple forms of resistance in microorganisms (including bacteria, viruses, parasites and fungi). As a result, although they were designed to heal, antimicrobials are becoming less and less effective and, ultimately, if no action is taken, there is a risk that they will no longer be able to protect us from infections.

Antimicrobial resistance would become responsible for almost 700,000 annual deaths worldwide2. If nothing changes, infectious diseases could become one of the leading causes of mortality worldwide by 2050, causing up to 10 million deaths2.
10 million Antimicrobial resistance would become responsible for almost 10 million deaths worldwide by 2050
The WHO is spearheading the international response to this scourge. Since 2015, it has organised World AMR Awareness Week, held on 18–24 November, which aims to increase awareness of this global phenomenon and encourage the general public, healthcare professionals and decision-makers to use antibiotics carefully, to prevent the further emergence of antimicrobial resistance.

As a major centre of expertise on the microbiota, the Microbiota Institute has been an active partner for the event since 2020. Throughout November, the Institute will be sharing articles and news, as well as expert videos, to help you understand the mid- and long-term effects of antibiotics on the human microbiota. To cite just one example, despite their well-known efficacy against bacteria (and lack of efficacy against viral infection3), antibiotics disrupt the balance of our gut microbiota. This imbalance, more commonly known as (sidenote: Dysbiosis Generally defined as an alteration in the composition and function of the microbiota caused by a combination of environmental and individual-specific factors. Levy M, Kolodziejczyk AA, Thaiss CA, et al. Dysbiosis and the immune system. Nat Rev Immunol. 2017;17(4):219-232. ) , is associated with several well-known problems, such as antibiotic-associated diarrhea. But there's more! Taking antibiotics is also suspected to increase the risk of multiple chronic diseases (allergies, asthma, obesity, chronic inflammatory bowel disease, etc.), particularly if they are prescribed in early childhood.

So can we do anything about it? Yes! Firstly, by ensuring that antibiotics are used sensibly and appropriately. Do not take these medications without a prescription from a healthcare professional. Stick to the specified dose, administration frequency and duration of treatment and do not share your antibiotics with anyone else4. And remember, as the French health slogan states:
If you are interested in the effects of antibiotics on your health and your microbiota, or if you want to know more about the World AMR Awareness Week (WAAW), we recommend that you go to this other dedicated page:
Each year, since 2015, the WHO organizes the World AMR Awareness Week (WAAW), which aims to increase awareness of global antimicrobial resistance.
Antimicrobial resistance occurs when bacteria, viruses, parasites and fungi change over time and no longer respond to medicines. As a result of drug resistance, antibiotics and other antimicrobial medicines become ineffective and infections become increasingly difficult or impossible to treat, increasing the risk of disease spread, severe illness and death.
Held on 18-24 November, this campaign encourages the general public, healthcare professionals and decision-makers to use antibiotics, antivirals, antifungals and antiparasitics carefully, to prevent the further emergence of antimicrobial resistance.
2. Tackling drug-resistant infections globally: final report and recommendations; May 2016.
3. Improving Antibiotic Use. Material Developed by CDC Using CDC materials does not imply endorsement or recommendation by CDC, ATSDR, HHS or the United States Government
4. Taking your Antibiotics. Material Developed by CDC Using CDC materials does not imply endorsement or recommendation by CDC, ATSDR, HHS or the United States Government

"Thanks for sharing!" -Gigi Snook (From My health, my microbiota)
"Nice 👍🏾" -Lucy Ofreneo (From My health, my microbiota)
"Interesting!" - Rémi Fresnel (From Biocodex Microbiota Institute on LinkedIn)
Is there a link between the microbiota (millions of bacteria) in your intestines and prostate cancer? It’s very likely, says a recent study published in Cancer Science.

A single purpose: explore the connection between prostate cancer and gut microbiota in a group of Japanese men. A single methodology: the profiles of the gut microbiota in men with and without high grade prostate cancer were compared. A single objective: determine whether the composition of the gut microbiota can be used as a new, non-invasive marker of high grade prostate cancer.
The cancer is diagnosed via rectal examination and a number of clinical examinations that assess the severity of the cancer and the risk of progression, and help to determine the
(sidenote:
Mohler JL, Antonarakis ES, Armstrong AJ, et al. Prostate Cancer, Version 2.2019, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2019;17(5):479-505.
)
. The vital screening and follow-up examination for prostate cancer is the prostate specific antigen (PSA) assay, although it does not accurately identify the grade of the cancer. The grade is assessed on a scale from 1 to 5, following examination of tissue samples from the prostate. At grade 1, patients’ prognosis is generally favourable, so it is recommended that no treatment be given, in order to avoid overtreatment which can often have an adverse impact. By contrast, patients with grade 2 prostate cancer and above require prompt, appropriate treatment. A new method for grading prostate cancers needs to be developed as a matter of priority to supplement the serum PSA test and avoid invasive examination.
In a previous study, the authors showed that in mice, obesity, a fatty diet and even certain molecules produced by the gut microbiota tend to promote the proliferation of prostate cancer cells. These results suggest that the gut microbiota could be used as a biomarker for determining how the cancer will progress. When they analysed the intestinal microbiota of patients who had undergone a prostate biopsy, the researchers found that three groups of bacteria were more abundant in patients with a high grade prostate cancer. To improve the accuracy of the diagnosis, the authors used a mathematical model and identified a further 18 types of bacteria to create the Fecal Microbiome Prostate Index, or FMPI. This FMPI index can be used to identify patients with a high grade prostate cancer with greater accuracy than conventional PSA assay.
While very encouraging, this study remains localised and the scope of the research is limited, with only men who were Japanese citizens being included. It now needs to be conducted in more patients, with different profiles, thereby confirming what are promising results.
Source:


