Scams, (fake) science, and publishing
The publication of scientific articles is essential to research but today this process faces two evils: predatory journals and doctored articles.
Details below.

The publication of scientific articles is essential to research but today this process faces two evils: predatory journals and doctored articles.
Details below.

The world of scientific publishing is essential to the sharing of research findings, but today it is plagued by two evils: predatory journals and paper mills.
Let’s take a closer look at this world without law.

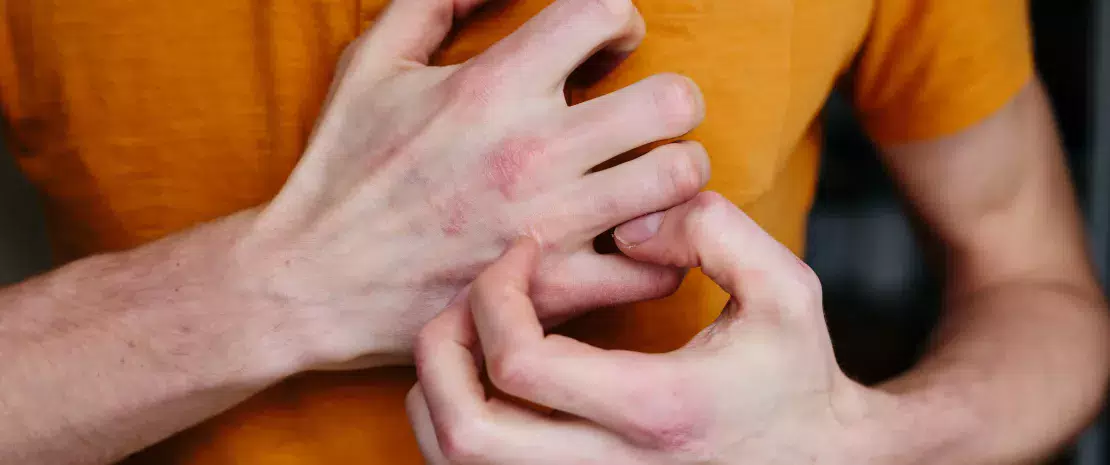
Atopic dermatitis (AD) is a chronic inflammatory skin disease that appears in periodic flareups. Like asthma, hay fever or allergic conjunctivitis, it is classified as an allergic disease. The disease causes very poorly defined oozing red lesions to appear in specific locations on the skin, such as in the folds of the elbow or behind the knees, but at times also on the face or the rest of the body. AD usually appears in early childhood, and may persist into adulthood. The causes are multifactorial and complex and include a genetic predisposition (mutation of the skin protein, filaggrin), an alteration of the skin barrier, a dysbiosis of the skin and gut microbiota, and immune dysregulation.
AD affects 15%-20% of children and 10% of adults in “developed” countries. The number of cases has increased significantly in recent decades due to pollution and contact with allergens.1
by Dr Genelle Healey
by Dr Genelle Healey
By Dr Genelle Healey
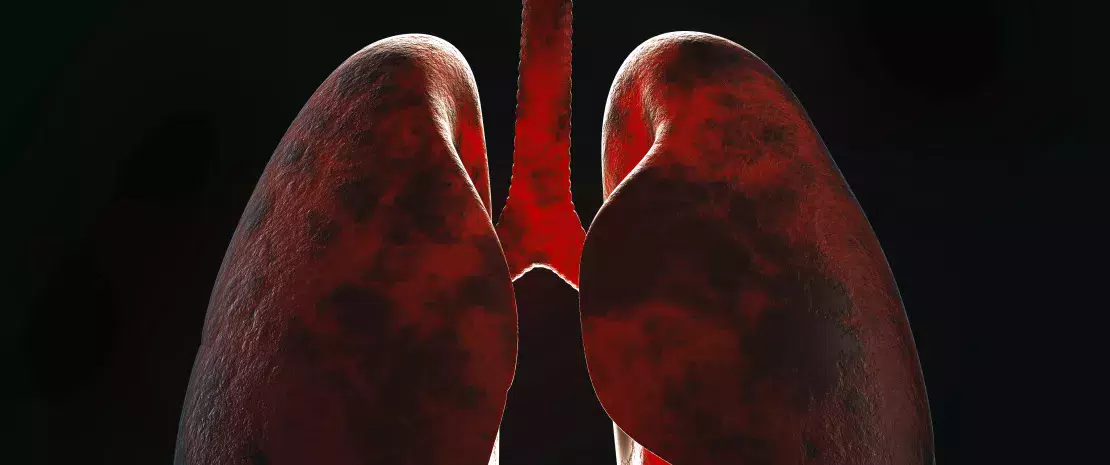
The microbiota plays a key role in the development, education and function of the immune system, both locally and systemically. While the airway microbiota locally regulates immune function, the gut microbiota can also influence respiratory immunity, via the gut-lung axis.1 Alteration of the lung and gut microbiota has been observed in many respiratory diseases, however whether the dysbiosis at these sites is a cause or a consequence of disease remains to be determined.2 Alteration of gut microbiota composition, through either diet, antibiotic use, aging, or disease, is associated with altered immune responses and homeostasis in the airways,3 highlighting that the gut microbiota can influence disease development throughout the body, including the risk of respiratory infections (Fig 6).4
