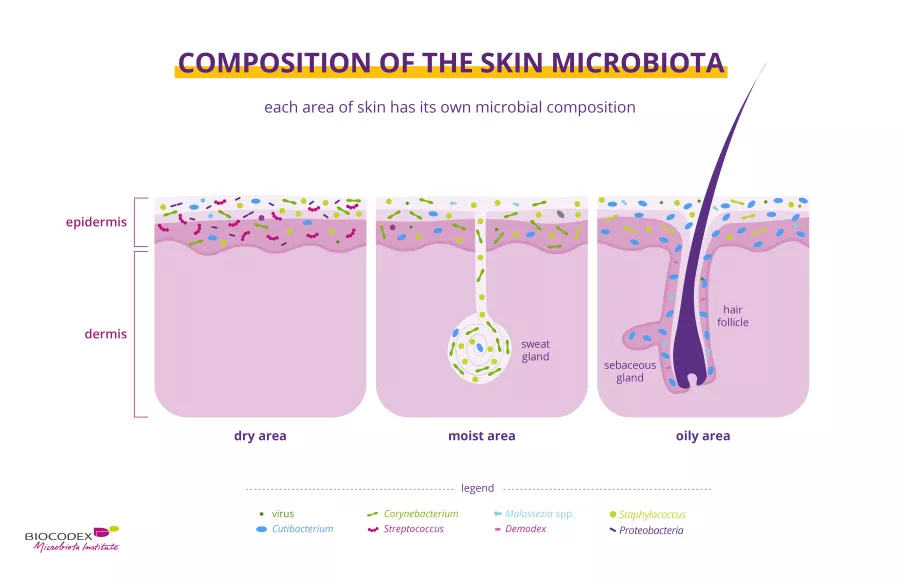
Obesity
Obesity is a worldwide problem that has tripled in the space of half a century.1 According to the World Health Organization (WHO), 39% of adults aged 18 and over are overweight and 13% are obese.
What if someone were to tell you that tucked away in our intestines, our gut microbiota could affect our appetite, our weight and our capacity for storing fat?2

What is obesity?
Obesity and overweight are most often the results of an imbalance between calories taken in and calories expended by the body – in particular, too much fat or sugar relative to actual energy expenditure.3,4 The resulting excessive increase in body fat can be harmful for health and significantly reduce life expectancy.3 Individuals who are overweight or obese are at greater risk of cardiovascular disease, as well as type 2 diabetes.5,6 Obesity is also linked to a number of cancers (such as liver and uterine cancer).6 Body mass index (BMI) provides an approximate indication of overweight and obesity. WHO considers overweight to equate to a BMI of 25 or more and obesity to a BMI of 30 or above.1
How does the microbiota fit in?
Genetic predisposition, a sedentary lifestyle, sleep deprivation, psychological factors and an unbalanced diet that includes too much fat and sugar are just some of the best known causes of obesity. Sometimes, however, adopting a wholesome lifestyle that combines exercise and a healthy diet is not enough to shed our excess weight.3 So, what's the reason? Recent studies of intestinal flora in obese patients have revealed disruptions in the composition of their gut microbiota, creating an imbalance in the microbial ecosystem defined as (sidenote: Dysbiosis Generally defined as an alteration in the composition and function of the microbiota caused by a combination of environmental and individual-specific factors. Levy M, Kolodziejczyk AA, Thaiss CA, et al. Dysbiosis and the immune system. Nat Rev Immunol. 2017;17(4):219-232. ) .7 Did you know? Rodent studies have shown that if the microbiota from an obese patient it transplanted into a mice, it gains weight as well!8 Surprised? These new insights offer hope for the discovery of new treatments. In fact, in obese individuals the microbiota is thought to be generally less abundant and less diverse,9 with fewer “good” bacteria such as Akkermansia muciniphila and bifidobacteria, and more potentially harmful bacteria.8 In such cases, the change in the balance of the microbiota is thought to allow the gut to extract energy from food more efficiently and therefore promotes energy storage in obese or overweight individuals.8
Repercussions beyond the gut
The balance between energy taken in and energy expended lies partly in bidirectional communication between the gut and the brain - what researchers refer to as the gut-brain axis.10 This communication between neurons and bacteria is achieved through “signal” molecules which include “ (sidenote: Short chain fatty acids (SGFA) Short chain fatty acids are a source of energy (fuel) for an individual’s cells. They interact with the immune system and are implicated in communication between the gut and the brain. Silva YP, Bernardi A, Frozza RL. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front Endocrinol (Lausanne). 2020;11:25. ) ” and “ (sidenote: Neurotransmitters Specific molecules that enable communication between the neurons (the nerve cells in the brain), as well as with the bacteria in the microbiota. They are produced by the individual’s cells and by the bacteria in the microbiota. Baj A, Moro E, Bistoletti M, Orlandi V, Crema F, Giaroni C. Glutamatergic Signaling Along The Microbiota-Gut-Brain Axis. Int J Mol Sci. 2019;20(6):1482. ) ”.5,6,11 The gut microbiota uses these messengers to help the brain to regulate energy balance, appetite and the feeling of satiety,6 and also modulates mood and eating behaviour.9,10 It can be seen that gut microbiota and obesity are inextricably linked; when the gut microbiota is impaired, it disrupts the digestion, the body’s defence system and the brain, with which it communicates to manage hunger.9 Like in a closed electrical circuit, the different “devices” are connected to one another and allow information (good or bad) to travel between the gut and the brain.
Can modifying the microbiota help to lose weight?
The solution ought to be straightforward, then: slim down and melt away those extra pounds by looking after your microbiota! The experts will tell you it's not quite that simple, though: it requires an understanding of how diet, probiotics and prebiotics, and faecal microbiota transplant (FMT) influence the ecosystem of our gut microbiota.
- Diet: Diet is of course the number one risk factor for obesity. It is also the main driver of microbiota modulation. Currently, no categorical link has been established between the effect on intestinal flora and the extent of weight loss.12 Conversely, the response to a diet is thought to be the result of the initial composition of our intestinal microbiota.7,11
- Probiotics: Many animal studies show that certain probiotics stand out and are thought to affect metabolic profile, weight gain or satiety in rodents.2,7,13,14 The results in humans are encouraging: while there is not so much data, certain specific probiotics had an impact on weight, BMI, waist measurement, body fat and metabolic profile.2,5,6
- Prebiotics: The results for prebiotics differ in humans, whereas their beneficial effects have been widely demonstrated in the laboratory setting2. Generally speaking, studies show that prebiotics have an effect on satiety,7 but that there is unfortunately no knock-on effect on weight.2
- Faecal microbiota transplant: Other therapies for modulating gut microbiota are being investigated, including faecal microbiota transplant (FMT), a current treatment for recurrent Clostridioides difficile infection. Researchers are evaluating its potential benefit in correcting dysbiosis and in influencing eating behaviour and energy metabolism.7
1 https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
3 Blüher M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019;15(5):288-298..
12 Seganfredo FB, Blume CA, Moehlecke M, et al. Weight-loss interventions and gut microbiota changes in overweight and obese patients: a systematic review. Obes Rev. 2017;18(8):832-851.
13 Lucas N, Legrand R, Deroissart C, et al. Hafnia alvei HA4597 Strain Reduces Food Intake and Body Weight Gain and Improves Body Composition, Glucose, and Lipid Metabolism in a Mouse Model of Hyperphagic Obesity. Microorganisms. 2019;8(1):35.
14 Legrand R, Lucas N, Dominique M, et al. Commensal Hafnia alvei strain reduces food intake and fat mass in obese mice-a new potential probiotic for appetite and body weight management. Int J Obes (Lond). 2020;44(5):1041-1051.