The pulmonary microbiota
Why is the lung microbiota so important for health?
Researchers long held the view that the lungs of healthy individuals were sterile.1 That's not the case, though! It's just 10 years since medicine discovered the abundance of lung microbiota.2 Unlocking its secrets since then has proved a major challenge.
What is the lung microbiota?
In contrast to its big brother, gut microbiota, it is only very recently that lung microbiota has been investigated. This delay can be partly explained by the difficulty of collecting samples and accessing the lungs themselves, both of which often require the use of an invasive method.3 In addition, the risk of bacterial infection of the upper airways is a real issue.4 Lung flora was first identified in 2010;5 there have since been very few studies devoted to elucidating the role of the communities of micro-organisms that live there.3
Nonetheless, our understanding of the composition of the lung microbiota can be summed up in three points:
- Low density of bacteria5
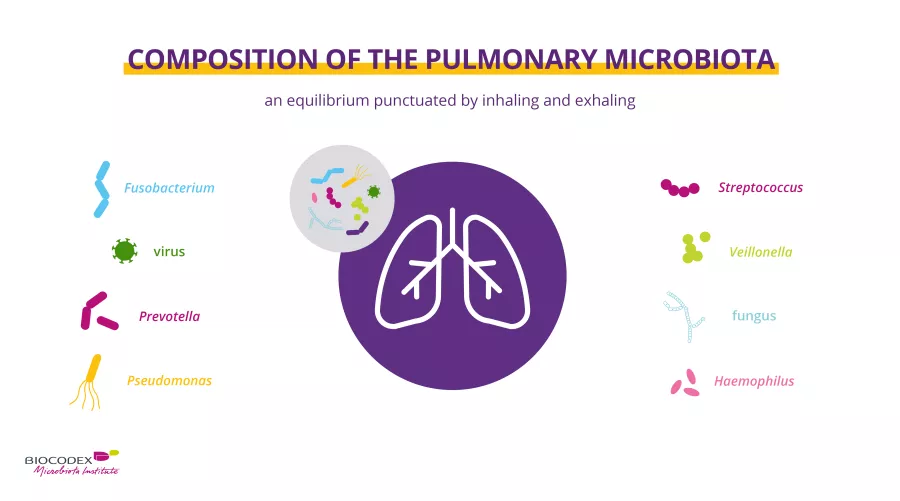
- High level of biodiversity. In healthy individuals, bacteria from the Prevotella, Veillonella, Streptococcus, Neisseria, Fusobacterium and Haemophilus5 genera are predominant in the lungs. Maintaining the balance of this bacteria relies on the rhythm of inhalation and exhalation6
- A tremendous abundance of “anaerobic” bacteria, which develop in the absence of oxygen. These are species of bacteria belonging to the Fusobacterium, Porphyromonas, Prevotella and Veillonella7 genera
Nor are bacteria the only (sidenote: Microorganisms Living organisms that are too small to be seen with the naked eye. They include bacteria, viruses, fungi, archaea and protozoa, and are commonly referred to as “microbes”. What is microbiology? Microbiology Society. ) that develop in the lung microbiota of healthy individuals! Fungi (species belonging to the Eremothecium, Systenostrema and Malassezia genera and to the Davidiellaceae family) and viruses (member species of the Anelloviridae family, along with a large number of (sidenote: Bacteriophage Virus that specifically targets and infects bacteria Scitable by Nature education_2014. Bacteriophage definition ) ) are also found to live there.2 Recent discoveries have also identified the presence of (sidenote: Archaea Type of micro-organism (different from bacteria) found in all environments, including extreme environments Archaea. Microbiology Society ) in the lungs.8 Lastly, while these micro-organisms form an integral part of the lung microbiota, the data currently available are limited.
Why does the lung microbiota play a major role in our health?
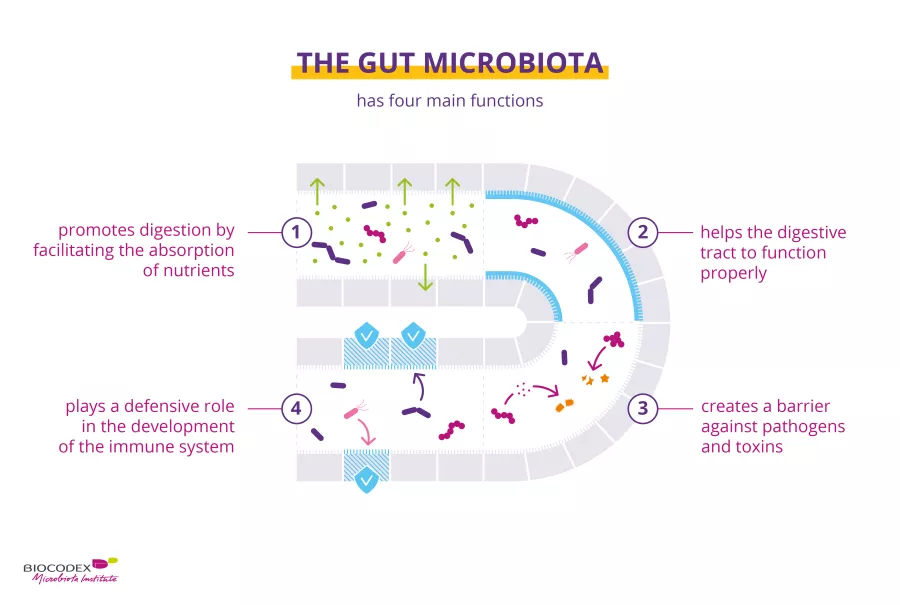
The lung microbiota plays a key role in maintaining the balance of lung function. It acts in four major ways:
- As a barrier to pathogens and an aid to resistance against respiratory infections,9 in exactly the same way as the gut microbiota
- It plays a defensive role by actively helping to stimulate (sidenote: Innate and adaptive immunity The human body protects itself using two kinds of defense mechanisms: innate immunity and adaptive immunity. Innate immunity is the first line of defense against disease agents and is an immediate response, while adaptive immunity is delayed but provides lasting protection Janeway CA Jr, Travers P, Walport M, et al. Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science; 2001. Principles of innate and adaptive immunity. ) in case of infections10,11
- It is involved in the maturation of the pulmonary immune system, enabling, inter alia, tolerance to allergens12
- It affects the morphology of the lungs and more specifically, the number of pulmonary alveoli, according to studies in mice13
Diseases linked to an imbalance in lung microbiota
When there is an imbalance in the composition of the lung microbiota, a (sidenote: Dysbiosis Generally defined as an alteration in the composition and function of the microbiota caused by a combination of environmental and individual-specific factors. Levy M, Kolodziejczyk AA, Thaiss CA, et al. Dysbiosis and the immune system. Nat Rev Immunol. 2017;17(4):219-232. ) 14 emerges, which may be linked to various diseases. Scientific studies to date have not always been able to determine whether the dysbiosis causes or results from such diseases, though.
Let’s take a closer look at the disorders linked to pulmonary dysbiosis:
- winter respiratory infections such as colds and flu (most often viral), where there is an imbalance in lung immunity, along with dysbiosis15 in the lungs and gut
- asthma:16 a chronic disease of the respiratory system affecting more than 260 million people worldwide.17 This condition is associated with an imbalance in the lung microbiota,16 as well as in gut18 and nasal19 microbiota
- cystic fibrosis, a rare genetic disease that mainly affects the airways and the digestive system20
- chronic obstructive pulmonary disease (COPD), which is characterised by progressive narrowing and permanent obstruction of the airways and lungs, causing difficulty breathing16
The “gut-lung axis”
These conditions highlight the bidirectional communication between the gut and the lungs, known as the “gut-lung axis”.15 Gut microbiota plays a role in pulmonary immune response and lung diseases can also influence the composition of the intestinal microbiota.15 In fact, many respiratory diseases go hand in hand with gastrointestinal disorders21 and, conversely, intestinal dysbiosis is often linked to lung diseases.16 The involvement of this axis in pulmonary diseases is a highly promising area for research.22
Looking after your lung microbiota
You now know about the important role played by the lung microbiota and the gut-lung axis in your health. Researchers have grasped this fully and are currently investigating strategies to prevent or cure lung infections.23,24
When it comes to preventing winter respiratory infections, there have been some encouraging results when combining probiotics with prebiotics.25 In infants, certain probiotics may well prevent winter infections as early as the first few months of life.26 Lastly, some studies in students have demonstrated the value of using probiotic treatment to achieve a significant reduction in symptom duration.27,28
All the information in this article is derived from accepted scientific sources. It should be remembered that this information is not exhaustive. The list of studies from which this information has been extracted can be found here.
17 World Health Organization. 2021. Asthma. World Health Organization, Geneva, Switzerland. https://www.who.int/news-room/fact-sheets/detail/asthm