Libido: gut bacteria spoiling the fun
Low libido is often blamed on excessive routine or the passing years. What if there were a less obvious culprit, one quietly nestled inside our gut?
The gut microbiota Diet
Low libido is often blamed on excessive routine or the passing years. What if there were a less obvious culprit, one quietly nestled inside our gut?
The gut microbiota Diet
Are structural and functional alterations in the gut microbiota responsible for muscle decline in the elderly (sarcopenia)? It seems more than likely, according to a recent study that looked at the largely unresearched gut-muscle axis in an elderly Chinese population. A promising avenue for improving health among the elderly?
The gut microbiota Alzheimer’s disease: the involvement of the oral microbiota has been confirmed Alzheimer’s: how gut dysbiosis influences amyloid pathology Rheumatoid arthritis: the gut microbiota is altered from the early stages
When faced with infection, the host produces taurine, a nutrient that feeds the microbiota and helps eliminate pathogens. As a result, taurine increases long-term resistance to subsequent infection.
The gut microbiota The gut microbiota has a specific signature for fibromyalgia Gut microbiota thought to block the effects of antidepressants Antibiotic exposure during first six years of life disrupts gut microbiota and impairs child growth
Loss of smell is a classic symptom of Covid-19. Particularly troubling for patients, this disorder is also a serious disability. Studies combining daily olfactory training and an analysis of the nasal microbiota are currently under way to help patients regain their sense of smell.
The ENT microbiota Diet
Markus Egert is Professor of Microbiology and Hygiene at Furtwangen University of Applied Sciences in Germany. His main research areas are the human microbiota (gut, skin) and the microbiota of the built environment. He studied biology and ecology and worked for four years in the consumer goods and cosmetics industry.
The skin microbiota Atopic dermatitis: nasal and skin microbiomes associated with disease severity Role of the microbiota in gut-brain communication Antibiotic exposure during first six years of life disrupts gut microbiota and impairs child growth
The existence of a gut-skin axis suggests the possibility of influencing the skin microbiota by modulating the gut microbiota. Pre- and probiotic oral solutions are therefore an option.
The skin microbiota Atopic dermatitis: nasal and skin microbiomes associated with disease severity Role of the microbiota in gut-brain communication Antibiotic exposure during first six years of life disrupts gut microbiota and impairs child growth
Preventing and treating dysbiosis without eliminating pathogens: novel therapeutic strategies aim to rebalance the skin microbiota directly via topical applications, or indirectly via oral solutions that modulate the gut ecosystem.
The skin microbiota Atopic dermatitis: nasal and skin microbiomes associated with disease severity Role of the microbiota in gut-brain communication Antibiotic exposure during first six years of life disrupts gut microbiota and impairs child growth
Staphylococcus epidermidis
Laced with blood vessels, packed with nerves, heavily involved in the immune system and massively colonized by microbial communities, the gut and the skin have a number of things in common.22 But that’s not all. Recent years have seen growing evidence for the existence of a link between the gut and the skin (the gut-skin axis) or even the gut-brain-skin axis.23
The skin microbiota Atopic dermatitis: nasal and skin microbiomes associated with disease severity Role of the microbiota in gut-brain communication Antibiotic exposure during first six years of life disrupts gut microbiota and impairs child growth

Human microbiome, conceptual image. Computer illustration showing the microbiome of the human body, including the colon (large intestine, lower centre). The microbiome is the collection of microbes (micro-organisms including bacteria, archaea, protists, fungi and viruses) found on and in the human body.
Changes in the skin microbiota can also be seen in non-pathological skin conditions. The skin is constantly exposed to various endogenous, exogenous and lifestyle factors that can affect the physical, mechanical or microbial properties of the skin barrier.19
The skin microbiota Atopic dermatitis: nasal and skin microbiomes associated with disease severity Role of the microbiota in gut-brain communication Antibiotic exposure during first six years of life disrupts gut microbiota and impairs child growth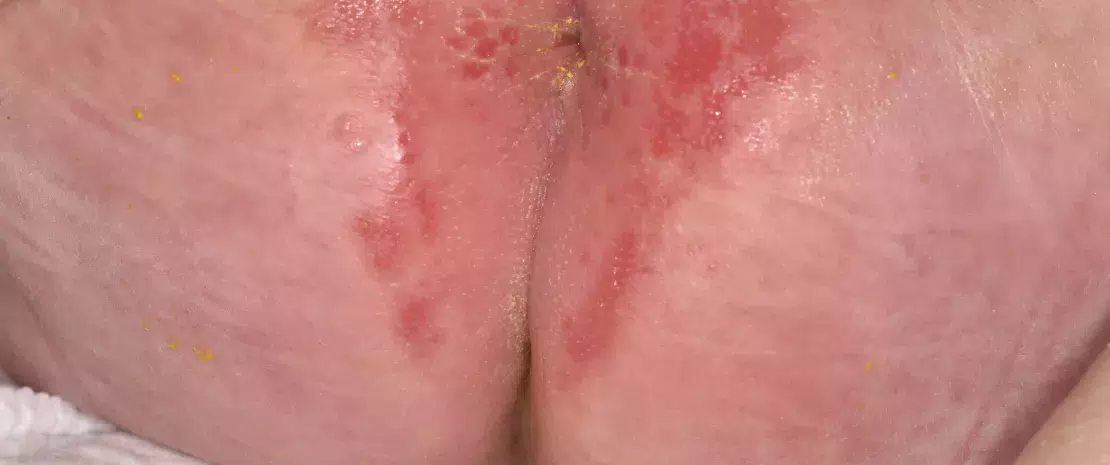
Nappy rash. Close-up of perianal dermatitis in a 1 month old female patient, showing red and inflamed skins.