Intestinal microbiota modulates tumour response in cancer patients
Press review
By Pr. Ener Cagri Dinleyici
Professor in Pediatrics, Eskisehir Osmangazi University Faculty of Medicine; Department of Pediatrics, Eskisehir, Turkey


Press review
By Pr. Ener Cagri Dinleyici
Professor in Pediatrics, Eskisehir Osmangazi University Faculty of Medicine; Department of Pediatrics, Eskisehir, Turkey

On February 4, 2018, World Cancer Day, the World Health Organization (WHO) stated on their website “Nearly every family in the world is touched by cancer, which is now responsible for almost one in six deaths globally. On World Cancer Day (February 4), the WHO highlights that cancer no longer needs to be a death sentence, as the capacity exists to reduce its burden and improve the survival and quality of life of people living with the disease” [1].
Over the last 10 years, tremendous advances have been made for cancer patients using new treatment strategies, including immune checkpoint inhibitors that target cytotoxic T-lymphocyte-associated antigen (CTLA-4) and programmed death 1 (PD-1) protein. However, therapeutic responses to these new treatment modalities are often heterogeneous, and some non-responder patients have been reported. The intestinal microbiome has been suggested to be an important host factor for non-responder patients, along with tumour genomics. Previous studies on microbiota and cancer have mainly focused on alterations in the intestinal microbiota of cancer patients (oncobiome) or microbiota precursors in order to define early-stage cancers, mainly colorectal cancers. However, promising new results regarding the influence of intestinal microbiota on anti-tumour immune responses have emerged. Two new studies were published in the first issue of Science this year.
• Gopalakrishnan et al. [2] evaluated the intestinal and oral microbiome in 112 patients with malignant melanoma, receiving anti-PD-1 immunotherapy, and compared baseline microbiota composition between cancer responders and non-responders. They revealed significant differences in the diversity and composition of intestinal microbiota between responders and non-responders. Significantly higher alpha diversity and relative abundance of Ruminococcaceae/Faecalibacterium was observed in responders, and this favourable intestinal microbiota composition has been suggested to enhance systemic and anti-tumour immunity among patients with melanoma. Patients with a low diversity and relatively high abundance of Bacteroidales (unfavourable intestinal microbiome) have impaired anti-tumour immune responses.
• Matson et al. [3] also evaluated the composition of baseline intestinal microbiota in patients with metastatic melanoma before receiving anti-PD-L1 therapy. Among the responders to treatment, Bifidobacterium longum, Collinsella aerofaciens, and Enterococcus faecium have been shown to be predominant members of the microbiota. These authors suggest that the commensal microbiome may exhibit a mechanistic impact on anti-tumour immunity in patients with metastatic melanoma. In the light of the results from these two previous clinical studies, it is thought that baseline intestinal microbiota may play a critical role in mediating the immune- stimulant response in melanoma patients receiving immunotherapy, such as anti-PD-L1 therapy. Further prospective studies are needed to reveal the precise interactions between the microbiome and cancer, not only in melanoma patients, but in terms of potential relevance for all types of cancer and the different treatment strategies.
1 World Cancer Day 2018. http://www.who.int/cancer/world-cancer- day/2018/en/
Congress review
By Dr. Julien Scanzi
Hepato-gastroenterology, Estaing University Hospital of Clermont-Ferrand and Thiers Hospital Centre, UMR INSERM/ UdA U1107 Neuro-Dol, Clermond-Ferrand Faculties of Medicine, France

The 7th GMFH summit was held in Rome on March 9-11, 2018. Once again, this year, internationally renowned physicians and researchers met to share the latest scientific advances in the field of microbiota; “a leading field of research”, as stated by Francisco Guarner, Chair of the Scientific Committee and leader of an ambitious programme.
The congress began with a Biocodex symposium on the impact of antibiotics on the gut microbiota. Dr. L. Armand- Lefevre recalled that antibiotics cause major alterations in the microbiota, in particular due to both the broad spectrum of antibiotics as well as high intestinal concentrations. In addition, microbiota resilience following antibiotic therapy may be slow and incomplete. In addition to the well-known short-term side effects, such as diarrhoea, taking antibiotics in early childhood is associated with an increased risk of obesity, allergies, and autoimmune diseases, as specified by Dr. A. Mosca.
Firstly, by trying to prescribe fewer antibiotics in a better way, moreover, if antibiotic prescription is necessary, by combining them with a probiotic. This is particularly the case for the probiotic, Saccharomyces boulardii, which limits dysbiosis and facilitates microbiota resilience following the discontinuation of antibiotics. Pr. C. Kelly has also shown that S. boulardii decreases the level of primary bile acids and increases that of secondary bile acids, thus reducing the risk of Clostridium difficile infection.
Our fibre consumption has decreased over recent decades, at least in the West, from more than 150 g per day a few generations ago to a dozen grams per day nowadays. This directly impacts the composition of our intestinal mucus. Pr. M. Desai’s Luxembourg team has shown, using a mouse model, that a low-fibre diet results in the intestinal mucus being more strongly degraded by the microbiota via glycoproteins contained in the mucus as an energy substrate. The resulting degraded mucus no longer plays its role against pathogenic bacteria such as Citrobacter rodentium, resulting in lethal colitis in these mice [1].
The potential role of the microbiota in colorectal carcinogenesis is well known. In a metagenomic study conducted in collaboration with Pr. J. Wang’s team in China, Dr. M. Arumugam demonstrated the existence of a“microbial signature” of colorectal cancer (CRC), based on the identification of four biomarkers which were significantly expressed in CRC patients compared to healthy subjects, in geographically different populations (China, Denmark, France, Austria). Of these biomarkers, two bacterial genes of Fusobacterium nucleatum (Fn) and Parvimonas Micra (Pm) were significantly over-expressed in cases of CRC [2]. Another recent study has confirmed the role of Fn as a biomarker of CRC, which significantly increased the sensitivity of immunological screening and made it possible to retrieve 75% of CRC cases which were negative on immunological testing [3].
With this advance in the recognition of a “microbial signature” of CRC, it may be possible to screen asymptomatic individuals for CRC in the near future based on an immunological test for blood in the stools coupled with microbiota analysis.
It has been known for a few years that the gut microbiota has an impact on the efficacy of chemotherapies. Recently, studies have also shown that the microbiota plays a major role in the response to immunotherapy. Based on a study of 26 metastatic melanoma patients, Pr. F. Carbonnel’s team showed that the type of microbiota is correlated with the response to ipilimumab (anti-CTLA-4). Patients with a microbiota rich in Faecalibacterium and other Firmicutes demonstrated a high response rate to ipilimumab and a significantly increased survival rate. The occurrence of ipilimumab-induced colitis was also more common in this group [4]. Similarly, another recent study based on 112 metastatic melanoma patients has shown that their responses to anti-PD-1 varied, and their microbiota, alpha-diversity, and relative abundance in Ruminococcaceae (a family whose main member is Faecalibacterium) were the main predictive factors for response [5].
As was the case last year, faecal microbiota transplantation (FMT) was the subject of a workshop and was often referred to in the various presentations. Drs. G. Ianiro and Z. Kassam recalled the very promising results of FMT for ulcerative colitis (two positive randomised controlled trials and one trial which showed a positive trend with FMT although significance was not reached), metabolic syndrome, hepatic encephalopathy, irritable bowel syndrome, and digestive GVH (graft-versus-host) allograft. Apart from recurrent Clostridium difficile infection, repeated FMT appears to be essential for “engraftment” and treatment efficacy. Administration in capsules seems to be the future for this technique but questions remain, in particular around intake dose and frequency, since these parameters are also likely to vary according to indication. Access to FMT is increasingly facilitated by the emergence of “stool banks”, in particular, in countries in which microbiota transplants have been assigned the status of organ/tissue rather than medicine. For example, in the United States, 98% of the population is within a two-hour drive of a centre practicing FMT. Thus, this practice has been widely used in recent years, but remains to be standardised and possibly adapted to patients according to their disease and microbiota.
Discovered in 2004, A. muciniphila is a bacterium that prevails in the mucus. It degrades mucin, stimulates butyrate production, and produces a pili-like protein, Amuc1100, that appears to play an important role in the immune response and barrier function of the intestinal mucus. It appears to have beneficial properties since its presence is inversely correlated to obesity, metabolic syndrome, and some cardiovascular diseases [6, 7]. In mice, its administration has beneficial effects on metabolic syndrome, and the first clinical data in humans should soon be available.
Congress review
By Dr. Aldo Maruy Saito
Paediatric gastroenterologist, Cayetano Heredia Hospital / Cayetano Heredia University in Peru, Lima, Peru

Dr. Ralijic-Stojanovic stressed that sequence analysis of 16S rRNA has made it possible to confirm that the taxonomy of many cultured intestinal microorganisms was incorrect. For example, Clostridium difficile, which does not belong to the Clostridium butyricum genus, is in fact a distant relative of Clostridium perfringens, in contrast to what was previously assumed [1]. Dr. Ralijic-Stojanovic recalled that between the ages of 7 and 12 years, the GM is still different to that in adults. He concluded by stressing that GM composition is individual, specific, and stable, and may vary depending on age, diet, and lifestyle.
In his presentation, Dr. Bäckhed reported that, while the role of the GM in metabolism is well known (optimisation of caloric availability, intake of enzymes absent in humans, and the role in vitamin K synthesis and short-chain fatty acid production), some more recent publications have revealed that the level of butyrateproducing bacteria is reduced in patients with type 2 diabetes, increased levels of Prevotella improve glucose metabolism, and Christensenellaceae bacteria may be considered as an anti-obesogenic probiotic [2]. Dr. Bäckhed concluded that the GM should be considered as an environmental factor that contributes to host physiology and metabolism.
The GM is very complex, and despite the advances in recent years, all its secrets are yet to be revealed.
The role of the GM in liver diseases is being increasingly understood, and some authors have even suggested the existence of a “gut-liver axis”. Dr. Gasbarrini discussed the role of GM in liver inflammation and fibrosis, showing that severe alterations in GM have been observed in cirrhotic patients, with increased levels of Enterobacteriaceae, Veillonellaceae, and Streptococcaceae, and decreased levels of Clostridiacea, Lachnospiraceae, and Eubacteriaceae. Dr. Gasbarrini believes that an insufficient resilience, resulting in adaptation through the acquisition of a dysbiotic microbiota, may contribute to the onset of GM-associated chronic diseases. Intestinal barrier breakdown is the cornerstone to progression of fibrosis and severity of liver cirrhosis.
Another interesting aspect was also discussed: depending on the mechanism underlying liver injury, the GM may induce or prevent hepatic fibrosis. Possible ways to restore a healthy GM include GM modulation (diet, rifaximin, probiotics or prebiotics) or “reinitialisation” through faecal microbiota transplantation.
Dr. Kobyliak presented a poster [3] on a study conducted in patients with non-alcoholic fatty liver disease (NAFLD), who received a probiotic combined with flaxseed oil and wheat germs or a placebo for eight weeks. The results show that the concomitant administration of probiotics and omega 3 reduces liver fat and serum lipid levels, improves the metabolic profile, and reduces the chronic inflammatory state. Dr. Kobyliak concluded that modulating the GM through the use of probiotics is a new option in the management of NAFLD.
This confirms the influence of the GM on liver diseases and the possibility of alternative solutions through the use of probiotics.
CIBDs are a heterogeneous group of immune-mediated chronic inflammatory diseases that affect the gastrointestinal tract. There are two main phenotypes of CIBD: ulcerative colitis (UC) and Crohn’s disease (CD). The relationship between the GM and CIBD is the subject of a growing number of publications.
Dr. Sokol discussed the pathogenesis of CIBDs and the fact that they are mediated by activation of the immune system through the GM in sensitive hosts under the influence of the environment. CIBD patients are known to have an abnormal microbiota with reduced diversity, which becomes increasingly reduced when the disease is active. Dr. Sokol stressed that there is an increase in Proteobacteria and a decrease in Firmicutes, which may or may not be correlated with the onset of disease. Thus, the level of adherent/invasive E. coli (Proteobacteria) is significantly increased in CD patients, but not in UC patients or healthy subjects. On the other hand, the level of Faecalibacterium prausnitzii (Firmicutes), which has antiinflammatory effects, is decreased in CIBD patients.
The environmental impact on GM is well known (mode of delivery, diet, antibiotics) and could also affect CIBD. Based on a report of a Danish cohort, Hviid et al. [4] observed a correlation between the number of antibiotic cycles received by a child and the risk of developing CIBD, which is greater for CD than UC.
Regarding pathogenesis, current controversy focuses on whether changes in the GM cause inflammation or vice versa; which came first: the chicken or the egg? Dr. Sokol believes that they are both at the same level, since the clinical manifestations of CIBD occur due to the implementation of a vicious circle between the GM and inflammation, and both may be the cause.
The data provided thus confirm the important role of GM bacteria in the pathogenesis of CIBD; however, our knowledge of the role of fungal GM in the pathogenesis of these diseases is limited. In this regard, Qiu et al. presented a poster [5] setting out the 15 main genera of fungi found in UC patients and healthy subjects (controls). For the Wickerhamomyces, Sterigmatomyces, and Penicillium genera, a positive correlation was observed with the expression of pro-inflammatory cytokines in the colonic mucosa, while the correlation was negative for Nigrospora. The authors concluded that the colonic fungal microbiota of UC patients is different to that of control subjects and that its alterations may be associated with mucosal inflammation and pathogenesis of UC.
Differences between the pathogenesis of CD and that of UC may be explained, in some cases, by the presence of a bacterial or fungal alteration of the GM.
3 Kobyliak T, et al. Co-administration of probiotic with omega-3 fatty acids in nafld management: evidence from animals to randomized clinical studies. OP343 N.
Commented article - children section
By Pr. Emmanuel Mas
Gastroenterology and Nutrition Department, Children’s Hospital, Toulouse, France
The mature human gut microbiota is established during the first years of life, and altered intestinal microbiomes have been associated with several human health disorders. Escherichia coli usually represents less than 1% of the human intestinal microbiome, whereas in cystic fibrosis (CF), greater than 50% relative abundance is common and correlates with intestinal inflammation and faecal fat malabsorption. Despite the proliferation of E. coli and other Proteobacteria in conditions involving chronic gastrointestinal tract inflammation, little is known about adaptation of specific characteristics associated with microbiota clonal expansion.
This study shows that E. coli isolated from faecal samples of young children with CF has adapted to growth on glycerol, a major component of faecal fat. E. coli isolates from different CF patients demonstrate an increased growth rate in the presence of glycerol compared with E. coli from healthy controls, and unrelated CF E. coli strains have independently acquired this growth trait. Furthermore, CF and control E. coli isolates have differential gene expression when grown in minimal media with glycerol as the sole carbon source. While CF isolates display a growth-promoting transcriptional profile, control isolates engage stress and stationary-phase programmes, which likely results in slower growth rates. These results indicate that there is selection of unique characteristics within the microbiome of individuals with CF, which could contribute to individual disease outcomes.[1]
The main digestive issue in cystic fibrosis (CF) is exocrine pancreatic insufficiency. This is present in 85% of cases and requires supplementation with pancreatic extracts. Despite this supplementation, fat malabsorption may persist. The proportion of Escherichia coli (E. coli) in the human gut microbiome is usually less than 1% but may reach up to 70-80% in patients with CF. There is clonal expansion within a given patient, but the strains are different between patients, which suggests an adaptation of E. coli to its environment. Some strains of E. coli are involved in gut inflammation and colorectal cancer (CRC). According to recent data, digestive inflammation, dysbiosis, or an increased risk of CRC may be associated with CF.
The authors have assumed that a selection of E. coli strains exist in patients with CF, as these strains are capable of surviving in a gut environment containing excess fat and abnormal mucus.
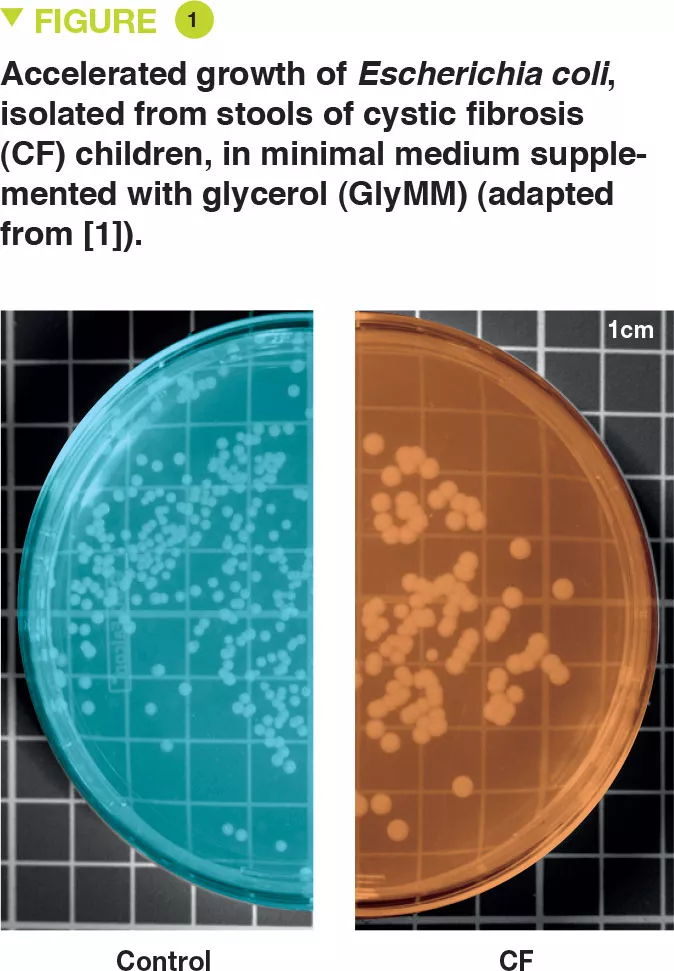
E. coli was isolated from the stools of six young children with CF and two controls. The authors assessed the growth of these bacteria in a minimal medium for which the only source of carbon was glucose (GluMM) or glycerol (GlyMM) supplementation. E. coli growth was faster in GlyMM plates for strains isolated from CF children compared to those isolated from controls (Figure 1). These differences were not observed under anaerobic conditions. Since the gut environment is essentially anaerobic, oxygen may play an important role, and in the vicinity of the epithelium, the oxygen gradient is minimal.
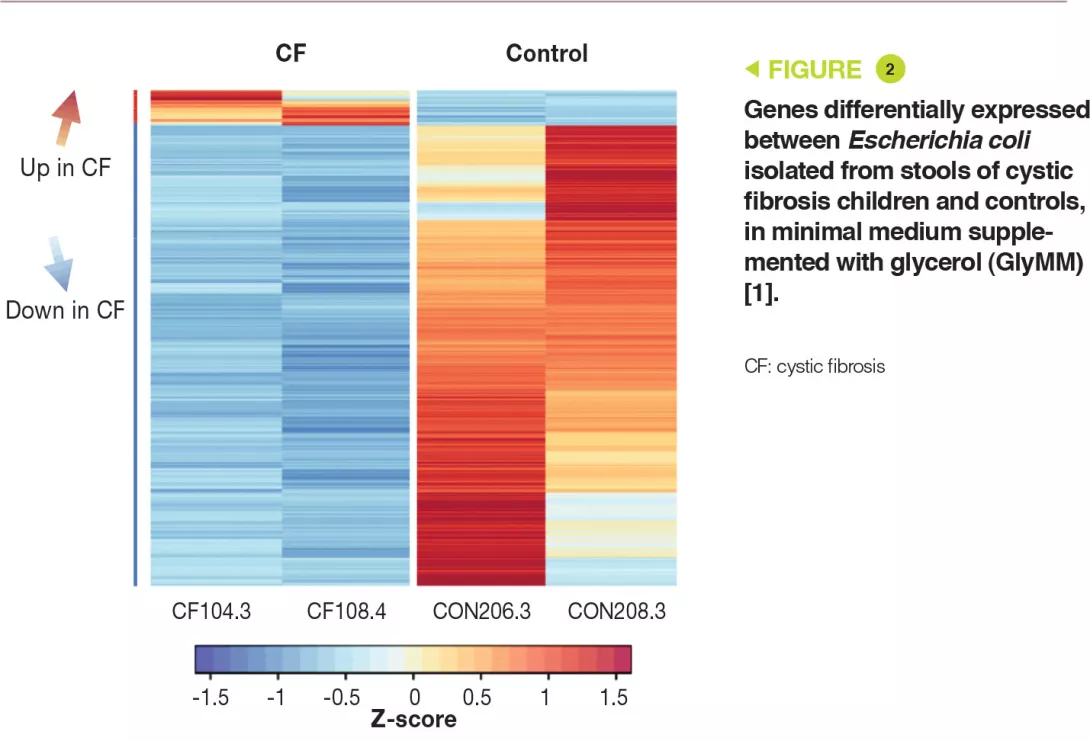
Genetic analysis has shown that the E. coli strains were distinct. In addition, the eight isolates had more than 11,000 SNPs (single nucleotide polymorphisms) present in one or more children with CF, which were absent in controls. Transcriptomic analysis (RNA-sequence analysis) in the GluMM or GlyMM medium was performed on isolates from two CF children and two controls. These isolates were selected due to their growth in GlyMM medium and their position within the phylogenetic tree. Among the genes expressed differentially, 213 were overexpressed (glycerol absorption and metabolism) and six were under-expressed (glucose cell transport) in GlyMM medium, in a similar way in both CF children and controls. In GluMM medium, only 20 genes were expressed differentially between CF children and controls, compared to 405 genes in GlyMM medium (377 genes were not induced in CF children) (Figure 2). The genes that were under-expressed in CF in GlyMM medium encode proteins involved in stress, acid resistance, and biofilm formation; the genes overexpressed in CF or under-expressed in controls encode proteins involved in growth mechanisms. In CF, the increased growth in GlyMM medium is not related to metabolic reprogramming, but to a loss of growth inhibition and a stress response.
Gut dysbiosis may be observed in CF. In particular, there is a significant increase in E. coli levels.
Due to fat malabsorption, intestinal glycerol levels are increased. In CF, selected strains of E. coli are adapted to these conditions with differentially expressed genes and a loss of growth inhibition.
This study leads to an understanding of the mechanisms involved in CF-associated dysbiosis with regards to E. coli. In order to correct this dysbiosis and limit, at least in part, gut inflammation, it is important to optimise the absorption of intestinal fat to ensure reduced glycerol levels.
An improvement in the intestinal barrier, especially with regards to mucus for this disease, may also reduce the availability of oxygen necessary for the growth of these E. coli strains.
In CF, the high intestinal level of glycerol due to fat malabsorption results in E. coli adaptation with clonal proliferation. Understanding these mechanisms may allow us to develop new therapeutic approaches and improve patient management.
Commented article - Adult section
By Pr. Harry Sokol
Gastroenterology and Nutrition, Saint-Antoine Hospital, Paris, France
Colorectal cancers (CRCs) comprise a complex mixture of malignant cells, nontransformed cells, and microorganisms. Fusobacterium nucleatum is one of the most prevalent bacterial species in CRC tissues.
Here, the authors show that colonisation of human CRC with Fusobacterium and its associated microbiome (including the Bacteroides, Selenomonas and Prevotella species) is maintained in distal metastases, demonstrating microbiome stability between paired primary and metastatic tumours. In situ hybridisation analysis revealed that Fusobacterium is mainly associated with cancer cells in metastatic lesions. In mouse xenografts of human primary colorectal adenocarcinomas, Fusobacterium and its associated microbiome remains viable despite successive passages. Treatment of CRC xenografted mice with metronidazole, an antibiotic, reduces Fusobacterium load, cancer cell proliferation, and overall tumour growth.
These observations should prompt further investigation of antimicrobial interventions as a potential treatment for patients with Fusobacterium-associated CRC [1].
Cancer-associated microbiota is known to influence cancer development and progression, including CRC. The microbiota of CRC patients is dysbiotic, and several studies conducted without any preconceptions have revealed an enrichment in F. nucleatum in tumour tissues and adenomas compared to non-cancerous colonic tissues [2]. These findings have been confirmed in studies conducted worldwide based on several cohorts of CRC patients. High levels of F. nucleatum have been associated with less T-cell tumour infiltration (T-cell infiltration is, however, a factor of good prognosis) [3], advanced disease, and a lower rate of patient survival. In addition, patients with F. nucleatum- associated CRC often presented with a right colonic location of cancer, BRAF mutation, and microsatellite instability. Studies based on various experimental models have suggested a protumourigenic role of Fusobacterium, which potentiates tumour growth in mouse models of CRC, xenografts derived from CRC cell lines, and in vitro CRC cell lines. The suggested mechanisms range from increased adhesion and invasion of tumour cells to modulation of the host immune response or activation of the Toll-like receptor 4 pathway. However, some animal or cellular studies have shown no carcinogenic effects of Fusobacterium [4].
To explore the role of Fusobacterium and its associated microbiota in CRC, the authors analysed the microbiota of five independent cohorts of CRC patients. For frozen samples (11 cases), the culture-based detection of Fusobacterium was positive in more than 70% of cases, showing that the bacterium was alive within the tumour tissue. Furthermore, when metastatic tissue was available and of good quality, Fusobacterium was also detected using culture methods. The molecular method (qPCR) demonstrated greater sensitivity, with more than 80% and more than 60% positivity for primary and metastatic tissues, respectively. The Fusobacterium identified in metastatic tissues was the same as that identified in the primary lesion. In addition to Fusobacterium, other bacteria with the same profile were identified, such as Bacteroides fragilis or Bacteroides thetaiotaomicron, however, unlike Fusobacterium, the strains identified in metastases were different from those identified in the primary lesion. In a cohort of 77 patients, the authors observed that there was no link between positive Fusobacterium culture and tumour recurrence. To determine whether the presence of Fusobacterium plays a role in carcinogenesis or is only passively involved in the oncogenic process, the authors used several systems, including human tumour cell xenografts in immunodeficient mice. They observed that Fusobacterium-positive tumours were easily implanted in mice while this was not the case for Fusobacterium-negative tumours. Finally, treatment with metronidazole, a highly active antibiotic targeting Fusobacterium, significantly reduced tumour growth (Figure 1).
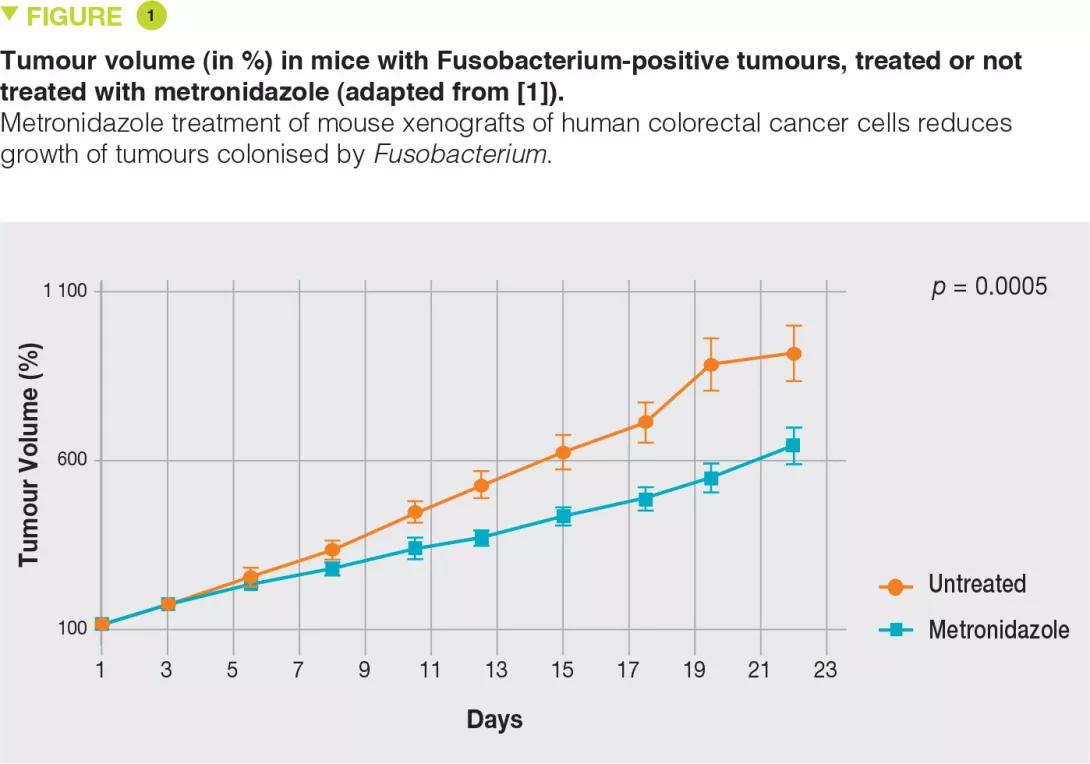
Fusobacterium is present in a viable form in primary and metastatic tissues in most CRC patients.
When present, Fusobacterium promotes tumour growth.
In murine models, antibiotic treatment targeting Fusobacterium significantly reduces the growth of Fusobacterium positive tumours.
The treatment of metastatic CRC remains a major clinical issue. This study shows that some bacteria of the microbiota, especially those belonging to the Fusobacterium genus, continue to be viable in both the primary tumour and metastatic tissues in most patients, and play a role in CRC progression. The use of antimicrobial treatments targeting these bacteria is therefore a strategy to be considered, while trying to be as specific as possible, because other bacteria may play a protective role or be involved in the response to conventional anticancer therapies and immunotherapies [5].
Based on several independant cohorts, this study shows that Fusobacterium is present in a viable form in primary and metastatic tissues in the majority of CRC patients, and is suggested to play a role in promoting tumour progression. These observations should prompt further investigation of antimicrobial interventions as a potential treatment for patients with Fusobacterium-associated CRC.
Overview
By Pr. Roberto Berni Canani, MD, PhD
Department of Translational Medical Science; European Laboratory for the Investigation of Food Induced Diseases, and CEINGE Advanced Biotechnologies; Task Force on Investigation on Microbiome, University of Naples “Federico II”, Naples, Italy
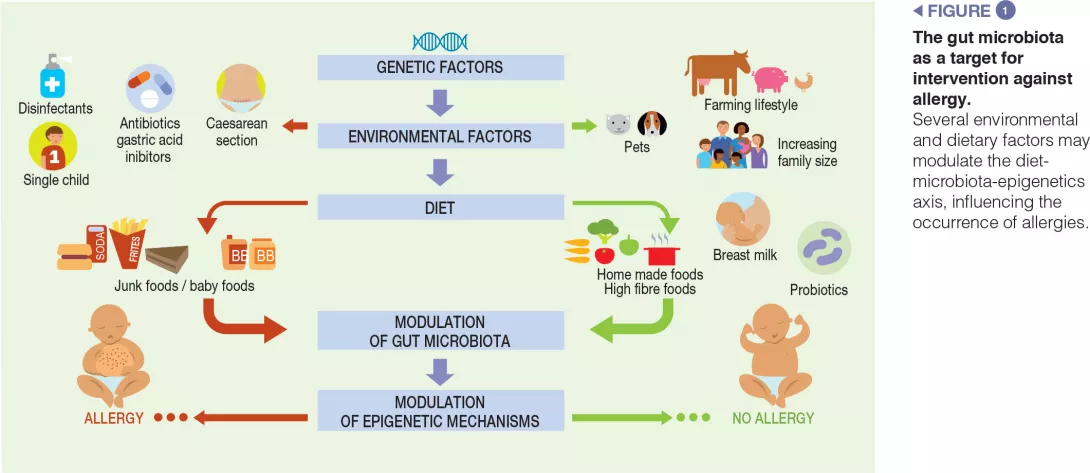
Dysbiotic gut microbiota plays an important role in the development of allergic diseases, in particular food allergies. The gut microbiota drives the maturation and function of the immune system, and genetic, environmental, and dietary factors may alter the commensal microbiota, leading to a dysregulation of immune function. Several factors responsible for dysbiosis have been associated with the occurrence of allergies, such as Caesarean delivery, the lack of breast milk, the use of drugs (mainly antibiotics and gastric acidity inhibitors), the use of antiseptic agents, and low-fibre/high-fat diets. No specific bacterial taxa have been consistently associated with allergies, but evidence suggests that gut dysbiosis occurs even before allergies present. Shortchain fatty acids (SCFAs) are crucial gut microbiota-derived metabolites involved in cross-talk with the immune system. Targeting the composition and function of the gut microbiota represents a promising strategy against allergic diseases, in particular, against childhood food allergies.
The prevalence, persistence, and severity of allergic diseases and, in particular, food allergies (FA), has increased substantially in recent decades in the industrialised world under the pressure of gene-environment interactions leading to immune system dysfunction, mediated, at least in part, by epigenetic mechanisms [1, 2]. This changing scenario has led to an increase in hospital admissions, medical visits, treatments, and a greater burden of care on families. All these factors have a significant impact on social costs and quality of life, and place a great psychological burden on patients and families.
Food allergies are characterised by an abnormal immune response towards dietary antigenic peptides that are normally tolerated. The cause of FAs is still largely undefined. Based on current knowledge, genetic susceptibility alone cannot account for the changing pattern of FAs and there has been a renewed interest in the role of the environment in the sensitisation to food. Evidence suggests a key pathogenetic role of gut microbiota (GM) alterations (dysbiosis) in allergy development. A healthy GM has a crucial impact on the development of the gastro-intestinal tract and immune system. A dysbiotic GM is associated with various diseases, including allergies [3].
The way in which dietary antigens are normally rendered non-immunogenic through immune tolerance has not yet been fully defined. Evidence suggests a pivotal role for regulatory T cells (Tregs) expressing the transcription factor Foxp3 (Foxp3+Tregs) and the complex interaction between the GM and immune and non-immune cells. The presence of both diet-and microbe-induced populations of Treg cells is required for full tolerance to food antigens [4].
During vaginal delivery, infants receive their first bacterial inoculum from the maternal vaginal tract, skin tissue, and often from faecal matter, exposing the immature immune system to a significant bacterial load [3]. The maturation of a healthy GM in early life allows for a change in the Th1/Th2 balance, favouring a Th1 cell response, while dysbiosis alters host-microbiota homeostasis, producing a shift in the Th1/Th2 cytokine balance towards a Th2 response [5].
Tregs are depleted in germ-free mice and in mice receiving an amino acid-based diet [4, 6]. Secretory IgA (sIgA) and innate immunity peptides exert a pivotal role in regulating GM composition. Deficiency in both innate and adaptive immunity (especially low levels of IgA) has been observed in children with multiple FAs [7]. A healthy GM promoting sIgA production facilitates the survival of protective bacterial strains within the gut lumen [8].
The expression of an allergic phenotype is dependent on the interaction between two major factors: genetic predisposition and gene-environment interactions. An increasing number of studies suggests a correlation between factors that alter the GM in the first years of life and the development of allergies later in life. There is increasing evidence that early-life GM dysbiosis represents a critical factor underlying the development of allergies.
The main factors responsible for dysbiosis are: birth by Caesarean section, lack of breast milk, drug use (mainly antibiotics and gastric acidity inhibitors), antiseptic agent use, timing of the introduction of solid foods, and junk food-based and/or low-fibre/high-fat diets [3, 9]. The maternal use of antibiotics before and during pregnancy, as well as antibiotic treatments during the first months of life, are also reported to be associated with an increased risk of cow’s milk allergies (CMA) in children [10]. Data that may be used to characterise the microbiota of patients with FAs are still preliminary. We recently described GM dysbiosis in children affected by IgE-mediated CMA; CMA infants had significantly reduced levels of Bifidobacteriaceae, Streptococcaceae, Enterobacteriaceae, and Enterococcaceae, and presented significantly higher levels of selected strains from Ruminococcaceae and Lachnospiraceae families. The GM of subjects with CMA comprised 73% Bacteroidetes and Firmicutes taxa, which are also known to dominate the adult gut [11].
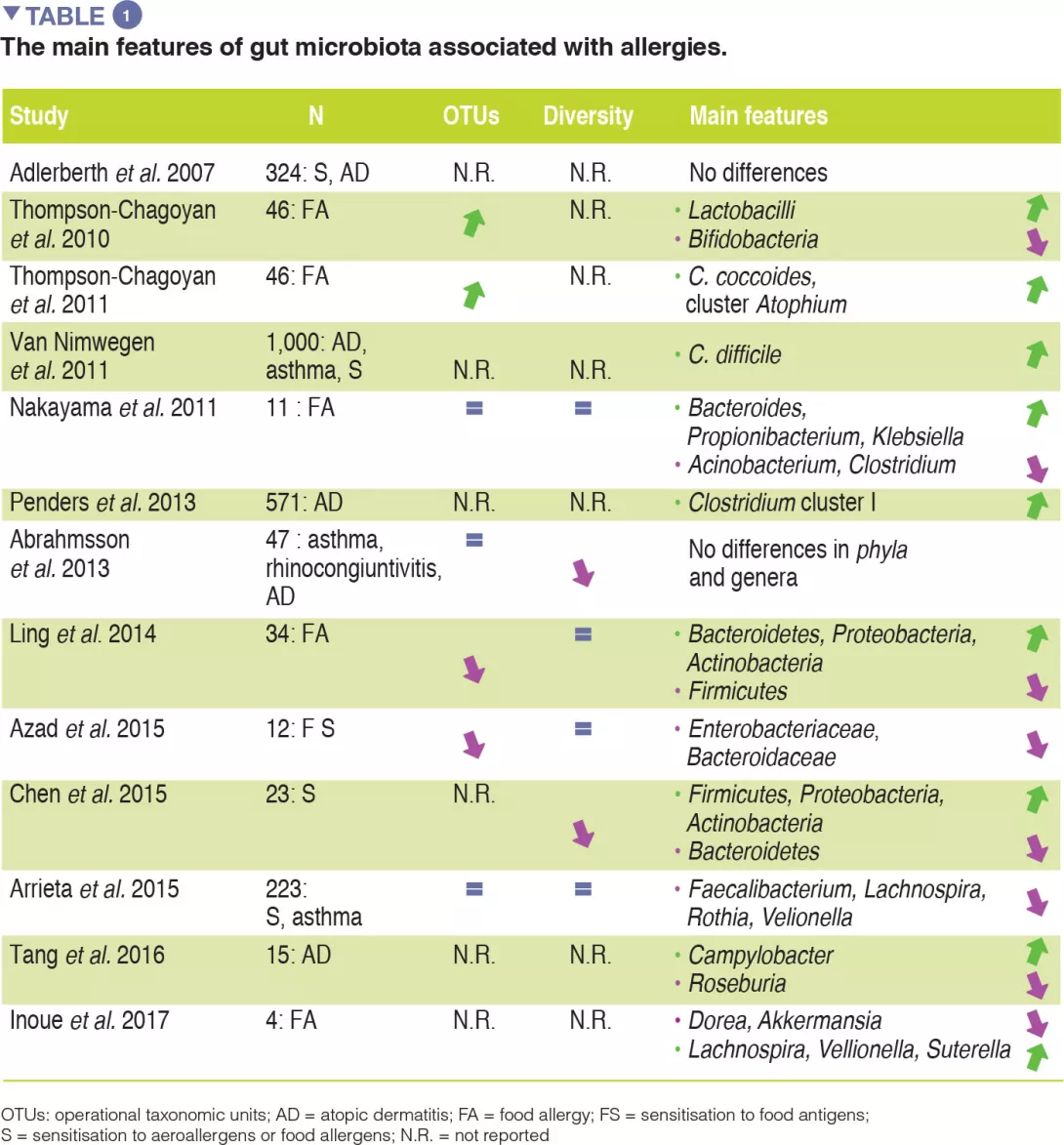
Although compelling evidence for an association between GM dysbiosis and FAs is emerging, heterogeneity in study design (involving sampling time points, the methods used to characterise microbiota, and allergic phenotypes studied) has made it difficult to establish a causal relationship between specific bacterial taxa and the development of allergies. No specific bacterial taxa have been consistently associated with FA and a broad range of microbes isolated from the human gut may be involved in tolerogenic mechanisms. The main evidence for allergy-associated GM is summarised in Table 1.
Evidence suggests that the gut microbiota-epigenetics axis, modulated by a number of environmental and dietary factors, plays an important role in the occurrence of allergies. This axis regulates a number of non-immune and immune tolerogenic mechanisms, and could be the ideal target for innovative, preventive, and therapeutic strategies against allergies.
SCFAs are major GM metabolites involved in cross-talk with human cells. Among the SCFAs, butyrate exerts a pivotal role in the induction of immune tolerance, and butyrate deficiency has been observed in allergic patients [4]. The different types of dysbiosis may be hypothesised to lead to similar effects in terms of SCFAs, and/ or the production of other microbiota-derived metabolites may facilitate the occurrence of allergies. Clostridia species, belonging to Cluster IV and XIVa, are the prominent source of SCFAs in the colon. Bacteria-produced SCFAs have been implicated in the regulation of both the proportion and functional capability of Tregs, which, in some studies, have been specifically attributed to butyrate production by spore-forming Clostridiales. An enrichment of taxa from the Clostridia class and Firmicutes phylum has been observed in human subjects with resolution of CMA [9]. Data from our laboratory showed that oral butyrate treatment leads to a dramatic inhibition of acute allergic skin response, anaphylactic symptom scores, reduced body temperature, increased intestinal permeability, and anti-βLG lactoglobulin (BLG) IgE, IL-4, and IL-10 production in a murine model of CMA, suggesting a protective role of butyrate against FAs [9]. Butyrate exhibits multiple mechanisms of action, however, many of these involve epigenetic regulation of gene expression through the inhibition of histone deacetylase (HDAC). The inhibition of HDAC9 and 6 increases FoxP3 gene expression, as well as the production and suppressive function of Tregs [12]. We evaluated the direct effects of butyrate on peripheral blood mononuclear cells (PBMCs) from children affected by challenge-proven IgE-mediated CMA. PBMCs were stimulated with BLG in the presence or absence of butyrate. Preliminary results show that butyrate stimulates IL-10 and IFN-γ production and decreases the rate of DNA methylation of these two cytokines. The same effective butyrate dose induces demethylation of the FoxP3 promoter region and down-regulation of HDAC6/HDAC9 expression [2].
Children exposed to farm environments have a lower risk of allergy development. Although it has not been conclusively proven, one of the plausible explanations for the protective effect associated with early- life farm exposure is the role of the GM, because individuals exposed to a farm environment possess a different microbial composition relative to those with other lifestyles [3]. Other epidemiological factors which protect against FAs include older siblings and exposure to pets in early life. Pet ownership is associated with high microbial diversity in the home environment. A recent study examining the influence of dietary patterns on the development of FAs at the age of two suggests that dietary habits may influence the development of FAs by changing the composition of the gut microbiota. In particular, an infant diet consisting of high levels of fruits, vegetables, and home-prepared foods was associated with less FAs [9] (Figure 1).
Probiotics, defined as ingested microbes that provide health benefits to the host, may be beneficial by modulating the GM [13]. Evidence of probiotic use against respiratory allergies remains preliminary (Table 2). However, meta-analyses have revealed that the use of selected probiotics, from gestation through to the first six months of life, could reduce the incidence of atopic eczema in children with a family history of allergic disease [14].
Emerging evidence supports the use of selected dietary strategies and probiotic strains for the prevention and treatment of allergies (Table 2).
We have previously demonstrated that addition of the probiotic, L. rhamnosus, to a hypoallergenic formula accelerates the acquisition of immune tolerance and protects against the occurrence of other atopic manifestations in children with CMA [15-17]. When we compared the faecal microbiota among infants receiving this tolerance-inducing probiotic therapy, we found significant positive correlations between the abundance of genera with a potential to produce butyrate and the concentration of faecal butyrate [11]. Strain-level demarcations for butyrate-producing genera (including Roseburia, Coprococcus, and Blautia), identified in infants who acquired tolerance to cows’ milk, suggest that L. rhamnosus treatment contributes to the acquisition of tolerance by altering the strain-level community structure of taxa with a potential to produce butyrate [11]. Accordingly, oral immunotherapy supplemented with another L. rhamnosus strain has been shown to be effective in inducing peanut non-responsiveness in 82% of allergic children [18].
The trillions of bacteria that populate our gut critically regulate key physiological functions against allergies. Environmentally induced changes in GM composition and function (decreased butyrate production, for example) create dysbiosis that is linked to an increased risk of allergy. Understanding how gut bacterial communities interact with the immune system is opening the way to novel preventive treatment strategies for allergies.
Press review
By Pr. Ener Cagri Dinleyici
Professor in Pediatrics, Eskisehir Osmangazi University Faculty of Medicine; Department of Pediatrics, Eskisehir, Turkey
Cystic fibrosis (CF) is a progressive, genetic disease that causes persistent lower respiratory infections and is related to different systemic symptoms and signs. More than 70,000 people live with CF worldwide [1].
The types and severity of symptoms can differ widely from person to person and are mainly related to the age of the patient as well as age at diagnosis. Children and adolescents with CF have a wide range of symptoms and signs including gastrointestinal manifestations. Recent studies have shown that dysbiosis is a feature of CF, leading to reports focusing on the relationship between the composition of the airway microbiota and clinical features and pulmonary function in patients with CF [2].
Dysbiosis-associated CF may be related to the natural course of the disease (including gastrointestinal involvement or respiratory microbiota alterations). However, patients require multiple courses of antibiotic treatment and these antibiotics may change the composition of the microbiota.
Studies have shown that patients with CF typically have decreased amounts of Bifidobacterium spp., Bacteroides-Prevotella group, Clostridium cluster XIVa, Faecalibacterium prausnitzii and Eubacterium rectale, whereas Enterobacteriaceae and Clostridia are increased. De Freitas and colleagues published a recent study in PLOS One, aimed at evaluating the effect of CF and antibiotic therapy on the intestinal microbiota composition in 19 children and adolescents with CF relative to 17 age and sex-matched controls [3]. The level of fecal calprotectin (an intestinal inflammation marker) was higher in the CF group (irrespective of antibiotic treatment) compared to the healthy controls. The authors showed that Bacteroides, Firmicutes, Eubacterium rectale and Faecalibacterium prausnitzii were significantly decreased, whereas Clostridium difficile, Escherichia coli and Pseudomonas aeruginosa were significantly increased in the CF group, relative to the healthy controls. The main differences in microbiota composition between patients with CF and controls, irrespective of antibiotic treatment, were noted for Eubacterium rectale, Bifidobacterium, Escherichia coli, Firmicutes, Pseudomonas aeruginosa and Clostridium difficile. The results of this study therefore demonstrate that the intestinal microbiota composition in CF patients is different from that of healthy controls and that the frequent use of antibiotics has no additional effects on these alterations.
Press review
By Pr. Ener Cagri Dinleyici
Professor in Pediatrics, Eskisehir Osmangazi University Faculty of Medicine; Department of Pediatrics, Eskisehir, Turkey

Most of the recent studies about the effects of drugs on the intestinal microbiota composition have focused on antibiotic use in different age groups.
Previous studies showed that metformin, proton pump inhibitors, NSAIDs, and atypical antipsychotics have an effect on the intestinal microbiota composition. However, these studies presented general results for drug classes instead of specific drugs. Lisa Maier and colleagues published their new study results in Nature in 2018, and they aimed to generate a comprehensive resource of more than 1,000 drugs actions on the microbiome (against 40 representative gut bacterial strains), which could facilitate more in-depth clinical and mechanistic studies, ultimately improving therapy and drug design. Maier et al. [1] showed that 24% out of these 1,000 drugs inhibited the growth of at least one strain in vitro. The effects of human-targeted drugs on gut bacteria are reflected on their antibiotic-like side effects in humans, like previously published human studies. This study showed that susceptibility to antibiotics and human- targeted drugs correlates across bacterial species, suggesting common resistance mechanisms, and highlighted the potential risk of non-antibiotics promoting antibiotic resistance. Widespread worldwide use of pharmaceutical drugs might be related with the dysbiosis, especially in modern Western societies.
This recent trial also showed that the intestinal microbiota composition can also modulate drug efficacy and toxicity and might be a new platform for further drug development; however, further in vivo clinical trials are necessary to better understand the mechanism of action. A comprehensive understanding of how therapeutics interact with gut microbes will open up the path for further mechanistic dissection of such interactions, and ultimately improve not only our understanding of the gut microbiome, but also drug safety and efficacy [2].
Congress review
By Dr. Jari Punkkinen
Head of Endoscopy Unit, Porvoo Hospital, Department of Medicine, Finland

Selected experts about Irritable Bowel Syndrome and Gut Microbiota.
Microbiota dysbiosis and its relation to irritable bowel syndrome (IBS) was discussed during various sessions of the conference. In particular, Pr. Magnus Simrén and Pr. Uday Ghoshal highlighted some features related to microbiota composition of IBS patients. Based on several studies, IBS patients have been shown to have a low microbial richness compared to healthy individuals, and Methanobacteriales may be undetected and methane production low in such patients [1].
Furthermore, a subset of IBS patients present dysbiosis with increased Firmicutes and Bacteroides enterotypes compared to healthy individuals with enriched Clostridiales and Prevotella enterotypes. However, an important question is not only which bacteria are associated with IBS but what they do in the gut and how they are involved in mechanisms of visceral hypersensitivity, neuro-motor dysfunction, increased permeability, and low-grade inflammation. Small intestine bacterial overgrowth (SIBO) may be the cause of IBS in some patients and a current challenge is to improve screening for these patients as upper gut aspirate culture is difficult to perform and not always available. The glucose hydrogen breath test may be used to identify such patients, and this would appear to be more accurate than the lactulose hydrogen breath test [2].
Can the gut microbiome be changed for therapeutic purposes and could it relieve IBS symptoms? Options to modulate gut microbiota include antibiotics, probiotics, synbiotics, altering gut motility, dietary manipulation, fecal transplantation, and use of bacteriophages. These therapies were discussed in the presentations of Pr. Uday Ghoshal and Pr. Giovanni Barbara. A role for antibiotics is most apparent in IBS-patients with SIBO. Both norfloxacin and rifaximin are significantly more effective in reducing IBS symptoms in SIBO- positive than SIBO-negative patients.
In IBS patients without constipation, according to Target 1 and 2 studies, rifaximin relieves global IBS-symptoms, and bloating. Target 3 and further studies have shown that rifaximin can be used repeatedly in relapsing IBS-D without loss of effect or appearance of bacterial resistance [3, 4]. Moreover, rifaximin transiently reduces bacterial counts in the feces but it also seems to have an eubiotic effect increasing Lactobacillaceae abundance.
The low FODMAP diet seems to reduce symptoms in some IBS patients but it also results in lower Bifidobacterium counts in the feces. In IBS patients responding to a low FODMAP diet the dysbiosis index increases, thus responsiveness to the diet may be predicted by faecal bacterial profiles. The efficacy of fecal microbial transplantation in IBS remains controversial as this was demonstrated in only one of the two large randomized controlled studies [5, 6]. As stated by Pr. Giovanni Barbara, the American College of Gastroenterology, based on a meta-analysis of 53 randomized controlled trials, concluded that probiotics reduce global IBS symptoms as well as bloating and flatulence [7]. To be consolidated, this recommendation should be founded on new, high-quality data.
However, not all probiotics are similar. Bifidobacterium infantis was shown by prof. Eamonn Quigley to relieve abdominal pain, bloating, and bowel function and improve quality of life in patients with all IBS-subtypes, and this appears to have anti-inflammatory and immunomodulating properties reducing CRP and TNFα in conditions like psoriasis, chronic fatigue syndrome, and ulcerative colitis. Furthermore, preliminary results suggest that in combination with Bifidobacterium longum it might also relieve depression in patients with IBS.
Based on preclinical studies, products of gut bacteria have been shown to change brain responses to stimuli, however the challenge is to translate these studies to clinical relevance. Huiying Wang showed in her recent study that Bifidobacterium longum strain modulates brain activity during social stress (associated with a cyberball game) in healthy volunteers based on evaluation using magnetoencephalography and QOL questionnaires. Besides an effect on neural oscillations, the strain also enhances a feeling of vitality and reduces mental fatigue compared to placebo over a four-week follow-up period. Pr. Paul Enck described the relationship between stress or anxiety and IBS as two-way as the symptoms can be both the cause and result of IBS. Based on a study of patients with IBS, Bifidobacterium longum was shown to correlate with a decrease in depression and anxiety scores, but at onset, these scores were insufficiently high to establish a diagnosis of depression or anxiety [8]. Thus, it is more appropriate to state that this probiotic affects mood rather than depression or anxiety. Similar to Bifidobacterium longum, rifaximin has also been shown to modulate brain activity and increase relaxation and reduce anxiety during social stress based on a double blindedrandomized trial of healthy volunteers evaluated by magnetoencephalography [9].
Congress review
By Dr. Solange Heller Rouassant
Pediatrician with specialty in Pediatric Gastroenterology and Nutrition México City, México Mexican Councilor of NASPGHAN

Proteobacteria, Firmicutes, Actinobacteria, Bacteroidetes, and Fusobacteria are the most abundant phyla in H. pylori positive and H.pylori-negative patients and this gastric microbiota may play a role in the H. pylori-associated carcinogenicity [1]. Alarcón [2] characterized gastric microbiota in children with and without H. pylori; when detected, H. pylori dominated the microbial community, but when absent, there was a higher bacterial richness and diversity.
Development of gut microbiota in early life is influenced by delivery mode, breast milk or formula feeding, antibiotic use, timing of introduction of solid foods and cessation of milk feeding. The gut microbiota of a newborn is transiently dominated by Enterobacteriaceae and Staphylococcus and very soon by Bifidobacterium and lactic acid bacteria. Bifidobacterium-dominated microbiota continues until complementary feeding starts [3].
Dukanovic [4] showed that C-section delivery and exclusive breast-feed infants have a relatively low abundance of Bacteroides in the infant stool; Bacteroides species were detected in 73% vaginal and 16% C-section samples.
Collado [4] demonstrated shared features between microbiota in maternal-infant pair and breast milk, suggesting a microbial transfer during lactation. Specific strains of Bacteroides, Bifidobacterium, Staphylococcus and Enterococcus genera were isolated from maternal infant gut and specific strains of Staphylococcus, Lactobacillus, Enterobacter and Acinetobacter from paired breast milk at 2 months of age.
It has been reported that association between gut microbiota composition in early life and development of diseases exists [5]. Studies on early infant gut microbiome have shown that antibiotic resistant gene carriage is acquired early and may have long term sequelae.
Exclusively breastfed infants were supplemented with Bifidobacterium longum subsp. infantis (Casaburi [4]) as a targeted probiotic capable of remodeling gut microbiome with potential reduction of antibiotic gene reservoirs. It was concluded that colonization of high levels of this strain is a safe and non-invasive method to decrease a reservoir of genes that confer antibiotic resistance.
High levels of Bifidobacterium longum subsp. infantis in breast-fed infants, regardless of delivery mode, remained stable through the first year of life if breastfeeding was continued [4].
Human milk oligosaccharides (HMOs) are unconjugated, solid and abundant compounds breast milk. The spectrum of HMOs in mother’s milk especially related to the mother’s secretor status modulates the bifidobacterial composition of the infant’s gut.
Gut of formula-fed infants has a lower relative abundance of Bifidobacteria and a higher microbial diversity. The use of prebiotics in infant formula increases the bifidobacterial fraction in the infant’s gut. Currently, available prebiotics (galacto- (GOS) and fructooligosaccharides (FOS)) are metabolized by Bifidobacteria, but not by the human host [5].
Puccio [6] supplemented an infant formula with 2 “fucosyllactose and lacto-N-neotetraose”, commonly found in human milk, with good results. An infant formula with GOS, FOS and Bifidobacterium breve compensates the delayed Bifidobacterium colonization in C-section-delivered infants, modulates gut microbiota and emulates conditions observed in vaginally born infants [6].
Comparison of two different infant formula supplemented with prebiotics only or prebiotics and probiotics showed similar gut microbiota profiles than breast-fed infants (Tims & Phavichir [4]).
Probiotics have been recommended for prevention of CMA even though more evidences are needed. Lactobacillus rhamnosus or Bifidobacterium lactis were administered daily from 35 weeks gestation to 6 months postpartum in mothers, and from birth until the age of two in children. Children that ingested Lactobacillus rhamnosus had a significant reduction of prevalence of eczema in childhood (Wickens [4]).
Extensive hydrolyzed infant formulas have been supplemented with L. rhamnosus for the management of IgE mediated CMA and development of immune tolerance. Clinical studies in healthy infants and infants suffering CMA showed that synbiotic-supplemented aminoacid-formulas (AAF) are hypoallergenic, well tolerated, and warranty normal growth.
Results of a multicenter, double blind, randomized controlled trial in infants with non IgE mediated CMA was presented (Candy [7]). Infants received a hypoallergenic, AAF formula containing a prebiotic blend of chicory-derived neutral oligofructose and long-chain Bifidobacterium breve . At week 8, significant differences on gut microbiota composition were present between groups, with higher percentages of Bifidobacteria in the symbiotic-AAF supplemented group. Modulation of gut microbiota using specific synbiotics may improve symptoms in infants with CMA.
Evidence suggests that altered gut microbiota affects gut motor function and induce gas production in infants, resulting in abdominal pain/colic. Manipulation of gut microbiota may play a role in the management and prevention of infantile colic.
