Vaginal Microbiota #21
By Pr. Satu Pekkala
Academy of Finland Research Fellow, Faculty of Sport and Health Sciences, University of Jyväskylä, Finland


By Pr. Satu Pekkala
Academy of Finland Research Fellow, Faculty of Sport and Health Sciences, University of Jyväskylä, Finland

Respiratory, gastrointestinal and neurodevelopmental complications: preterm birth is the main cause of neonatal morbidity and mortality. The vaginal microbiota seems to be involved, but the underlying mechanisms remain poorly understood. A team of American researchers tracked the genome of the vaginal microbiota of 175 American women throughout their pregnancies (40 of whom subsequently experienced spontaneous preterm delivery, and 135 of whom delivered at full term). The study shows that the two types of pregnancy differ in terms of vaginal microbiota composition: certain bacterial species of the Lactobacillus genus, such as L. helveticus, L. crispatus, L. gasseri and L. jensenii, are associated with fullterm pregnancies, while Megasphaera genomosp, Gardnerella spp. and Atopobium vaginae are linked to preterm births. Another finding is that the genetic diversity of the vaginal microbiota is higher in the first half of pregnancies that end preterm, due to Gardnerella species. More precisely, the nucleotide diversity of Gardnerella spp. increases at the start of pregnancies that end preterm whereas it remains stable in pregnancies that are carried to term. The genetic diversity of Gardnerella spp. could perhaps be used as a biomarker for the early diagnosis of preterm birth. But how can we explain this peak in Gardnerella nucleotide diversity? Compared to other bacteria, Gardnerella shows a 1.5-fold higher growth rate at the start of pregnancy, more frequent genetic recombination and greater selection of mutations that benefit this bacterium (and increased elimination of deleterious mutations). Antibiotics and other xenobiotics are thought to be involved. In fact, the more diversified gene pool of G. swidsinskii seems to correspond to an adaptation to drugs, confirming a previously suggested effect of xenobiotics in the vaginal environment; and vaginal microbiota associated with preterm birth exhibit higher antibiotic resistance potential. Genomic variation in vaginal bacteria is therefore believed to affect the host’s phenotypes (including pregnancy outcomes). However, the authors do not rule out another explanation, even if they consider it unlikely: the associations between microbial genetic diversity and pregnancy outcomes could also result from unmeasured confounding factors (drugs, chemical compounds, etc.) that might act on both variables.
By Pr. Satu Pekkala
Academy of Finland Research Fellow, Faculty of Sport and Health Sciences, University of Jyväskylä, Finland

Severe acute pancreatitis (AP) patients are at risk of elevated mortality, for which determining the course of disease within the first few hours would be very important. The current complex scoring systems cannot predict AP severity early enough, and thus, novel markers are needed. While there seem to be a bilateral link between AP and gut microbiome, larger prospective clinical studies have been lacking. This paper presents results of orointestinal microbiome from 450 patients with AP from 15 European centers. The samples were sequenced by full-length 16S rRNA and metagenomic sequencing using Oxford Nanopore. The revised Atlanta classification (RAC) redefines severity of AP into three categories: mild, moderate, and severe (RAC I-III, respectively). This study found that Bray-Curtis distance of the rectal microbiomes was different in RAC III compared with RAC I and RAC II. Further, several bacterial species were differentially abundant depending on the RAC category. Bray-Curtis distances were also different between alive and deceased patients in rectal but not buccal microbiomes. In addition to mortality, the length of hospital stay associated with early alterations of rectal microbiome. In the end, the authors found that 16 bacterial species were differentially abundant in severe vs. non-severe AP. In Ridge regression, these species together with systemic inflammatory response syndrome could faithfully predict disease severity. Interestingly, all these species are producers of short-chain fatty acids (SCFA). Accordingly, functional pathways of SCFA production were more expressed in severe AP. While the finding is intriguing, it is still unknown whether SCFA producing bacteria are cause or consequence of severe AP.

The incidence of type 2 diabetes (T2D) is increasing drastically in Middle East countries. Several Western studies have shown the contribution of gut microbiome in T2D-associated insulin resistance and low-grade inflammation, but studies in Middle East populations are scarce. Further, the existing studies show inconclusive results of how the microbial community composition and functions contribute to the pathogenesis of T2D. To gain more insight, the authors analyzed stool samples of 84 individuals from the United Arab Emirates with or without T2D using nanopore metagenomic sequencing. Unlike many earlier Western studies, this study reported no differences in gut microbiota alpha-diversity between healthy controls and T2D. Further, after correcting for multiple comparisons, the authors did not find differential abundance of any microbial species or KEGG orthology (KO) features between the groups. However, a gene set enrichment analysis revealed 8 functions with higher abundance in the control group and 5 in the T2D group. These differentially abundant modules associated with the degradation of amino acids, such as arginine, the degradation of urea and homoacetogenesis. These functions seem to have pro-inflammatory effects, and thus, may contribute to low-grade inflammation, a hallmark of T2D. Ultimately, the authors used prediction analysis to identify 3 potential biomarkers of T2D. These included a depletion of Enterococcus faecium and Blautia as well as an enrichment of Absiella spp or Eubacterium limosum in T2D. Interestingly, E. faecium is shown to have lipid-lowering and anti-obesity effects, and therefore, might partly contribute to the pathogenic T2D phenotype. To conclude, this study was successful in identifying specific microbial biomarkers, including functions and taxa that may help in predicting the development of specific T2D-associated disease conditions.

Gastric cancer (GC) is one of the leading causes of cancer death worldwide. Early detection is important for successful treatment of GC. Programmed death-ligand 1 (PD-L1), a target of cancer immunotherapy, is highly expressed in tumor-associated macrophages that can be regulated by the gut microbiome. One possible way through which the microbiome may have anti-cancer effects is the production of short-chain fatty acids, including butyrate. In this study, advanced GC patients expressed more immunosuppressive markers, namely PD-L1 and interleukin (IL)-10, in macrophages, dendritic cells and cancer mucosa than healthy controls. The gut microbiota of the GC patients was characterized by lower diversity and dysbiosis. At genus level, lower abundances of butyrate-producing bacteria, such as Faecalibacterium and Bifidobacterium were detected in GC patients. Interestingly, administration of butyrate and Faecalibacterium into the peripheral blood mononuclear cells of GC patients decreased the number of PDL1- and IL-10-expressing macrophages. In addition, butyrate suppressed the growth on cultured GC cells. However, it remained unclear which Faecalibacterium strain was used in the in vitro experiment. Ultimately, a humanized tumor mouse model was injected with GC cells and peripheral blood mononuclear cells of from healthy controls or GC patients with or without butyrate. The experiment showed that butyrate significantly decreased tumor size and the immunosuppressive markers PD-L1 and IL-10. Thus, butyrate may have therapeutic potential via suppressing cancer cell growth in GC.
By A/Pr. Dao Viet Hang, MD, PhD
Hanoi Medical University, Vietnam

In December 2023, more than 3,000 physicians representing over 60 countries attended the Asia Pacific Digestive Week in Bangkok. The event provided a rich and diverse program, with many updated lectures spanning various fields including hepatology, endoscopy, gastrointestinal (GI) diseases, and motility/surgery. Besides the lectures on newly released guidelines, many new topics are integrated into the program with various formats – interactive sessions with case-based discussion, debate sessions on different aspects and approaches, and keynote lectures from well-known panellists worldwide.
One of the highlights of this year’s event are numerous sessions dedicated to GI motility, with a main focus on functional diseases. In the opening ceremony, an update on refractory gastroesophageal reflux disease (GERD) was presented as a presidential lecture from Prof Somchai Leelakusolvong, President of Local Organizing Committee. Prof Somchai emphasized the importance of the Lyon consensus version 2.0, which has expanded the criteria of endoscopic findings to include Los Angeles reflux esophagitis grade B, which is more practical in Asian countries. The event also introduced many updated data on optimizing the treatment of refractory GERD based on various mechanisms. The advancements in treatment strategies were also highlighted, including the use of drugs targeting lower esophageal sphincter (LES) pressure, esophageal contractions, endoscopic interventions, and electrical stimulation. Transient lower esophageal sphincter relaxations were considered as one of the key mechanisms of GERD. This condition can be improved by baclofen by increasing resting LES pressure, thus reducing episodes of reflux. Preliminary data on a small cohort of patients suggested that electric stimulation could improve LES pressure; however, the practical application of this intervention in the future is still debated.
The event also paid considerable attention to the comparison between proton pump inhibitors (PPI) and potassium‐competitive acid blockers (PCAB) in different studies, with the target population being patients with erosive esophagitis. Current evidence showed a higher efficacy of PCAB compared to PPI in treating severe erosive esophagitis with acceptable adverse events.
One of the most engaging sessions was “All about GERD”, chaired by Prof Somchai Leelakusolvong and Prof Kwang-Jae Lee on December 8th. This session primarily focused on the updates of the modern Lyon consensus, non-acid reflux management, and optimizing treatment of functional heartburn.
Dr. Ping-Huei Tseng, Taiwan, presented the detailed changes of the Lyon consensus 2.0 with the clarification on the expanded criteria in endoscopic findings for Los Angeles grade B esophagitis. The role of high-resolution manometry to exclude mimic esophageal disorders and identify risk factors of GERD such as low LES pressure, hiatal hernia, or weak oesophageal contraction was also explained with case examples for further clarity. Some promising metrics on 24-hour pH impedance, such as mean nocturnal baseline impedance (MNBI) and post-reflux swallow-induced peristalsis (PPSW) index, are still debating and require further clinical data.
For non-acid reflux management, Prof. Justin Wu from Hong Kong highlighted the differences between the definition of refractory GERD and refractory GERD symptoms, of which the latter can be caused by various diseases. The roles of high-resolution manometry (HRM), endoscopy, and 24-hour pH impedance in the diagnosis and management of these conditions are explained in detail by the ESNM/ASNM guideline. The decision to perform 24-hour pH impedance on or off PPI depends on the diagnostic aim, whether to confirm GERD in patients with no prior diagnosis, or to confirm refractory GERD. It will be helpful to have a stepwise strategy for patients with refractory GERD to determine the optimal time for endoscopic interventions or surgery. Non-acid reflux management should be considered comprehensively for possible mechanisms, including characteristics of reflux episodes, oesophageal motility patterns, and overlapping symptoms. Furthermore, Prof Wu emphasized the need to establish a cut-off value for acid exposure time (AET) in GERD diagnosis for the Asian population, which can be a debating point when compared to the Lyon consensus.
Functional heartburn is also a challenging condition due to several factors: overlapping with other functional gastrointestinal disorders, presenting with mental disorders (anxiety, depression, stress) in the scope of the “gut-brain pathway” mechanism, and requiring exploration tests for exclusion. According to recent data, 70% of patients with functional symptoms had normal endoscopic findings. Within this population, 50% had normal 24-hour pH impedance results, and 60% showed no correlation with the occurrence of symptoms, meaning only 21% was classified as functional heartburn. That is why, besides PPI, neuromodulators play an essential role. Tricyclic antidepressants (TCAs) and selective serotonin uptake inhibitors (SSRIs) have shown efficacy in treating functional heartburn. However, their potential side effects should be carefully considered. For prevention, it is recommended to initiate treatment with a low dose and maintain follow-up during treatment.
Artificial intelligence (AI) is also a hot topic with many invited speakers. On the final day of APDW, December 9th, ASPDE cohosted with WEO to organize a session called “ASPDE-WEO International Clinical Symposium Artificial Intelligence in Endoscopy: Implementation in the Asia Pacific and the World”. This session was moderated by Prof Hisao Tajiri, Prof Yuichi Mori, and Assoc. Prof Nonthalee Pausawasdi. Prof Yuichi Mori gave the first presentation to introduce the WEO AI committee to the two ongoing projects. One project is an international study aimed at evaluating the perceptions of endoscopists and patients regarding the use of AI in endoscopy. The other is a longitudinal study on the role of AI in real-world settings. The WEO AI committee focuses on implementing AI in clinical practice, considering different aspects including accuracy, cost-effectiveness, doctor-machine interactions, training programs, and ethical considerations.
Prof Han-Mo Chiu, Prof Rungsun Rerknimitr, and Prof Kherk-Yu (Lawrence) Ho each presented different topics on developing and using AI effectively in several fields, including colorectal cancer screening, gastric cancer screening, and biliary endoscopy. The presentations showed many updated data, inspiring clinicians and endoscopists to consider implementing AI in the near future.
Assoc. Prof Dao Viet Hang presented another aspect of utilizing AI in endoscopy training, especially in limited-resources settings. She highlighted that the conventional metrics in endoscopy training, based on the minimum number of cases or the duration of practice, do not reflect the skills and personal development over time, requiring a more interactive approach. E-learning training programs and simulation-integrated activities have shown promising results in enhancing junior endoscopists’ knowledge and lesion detection skills. Until now, AI has shown promising data on improving lesion detection with more and more data in clinical practice; however, its integration in endoscopy training is still lacking. Some key considerations for applying AI in endoscopy training include economic feasibility, safety and accountability, technical concerns and validation, and clinicians’ role in digitalization. The framework suggested that adopting AI in endoscopy training should balance users’ factors, technology factors, social factors, and contextual factors (educational environment and standards). A needs assessment is required to outline educational needs and establish clear educational goals to inform Al’s technology selection. Al should be integrated into training based on the best evidence and within a curriculum, incorporating user training for both trainees and trainers to promote uptake.
All the talks in this session received substantial feedback, comments, and questions, reflecting a great interest in the future application of AI in endoscopy.
Viviani Mendes de Almeida
Laboratory of Microbiota and Immunomodulation - Department of Biochemistry and Immunology, Institute of Biological Sciences, Universidade Federal de Minas Gerais - UFMG, Belo Horizonte, Brazil
Angélica Thomaz Vieira
Laboratory of Microbiota and Immunomodulation - Department of Biochemistry and Immunology, Institute of Biological Sciences, Universidade Federal de Minas Gerais - UFMG, Belo Horizonte, Brazil
Daiane Fátima Engel
Department of Clinical Analysis, School of Pharmacy, Universidade Federal de Ouro Preto - UFOP, Ouro Preto, Brazil and Center for Social and Affective Neuroscience, Linköping University, Linköping Sweden

is a PhD student under Pr. Angélica Thomaz Vieira’s supervision. Viviani Mendes was selected from the special call of paper of the Microbiota Mag. She gives us a tour from her recent publication about the influence of microbiota in post-Covid effects. Her study was recently published in Gut Microbes [1].
Covid-19 has wreaked havoc on a global scale, resulting in millions of confirmed cases and fatalities as of March 2023. Long-term complications of Covid-19 are pervasive, affecting even individuals with mild or asymptomatic cases. Among pathophysiological responses triggered by Sars-CoV-2 infection, several studies have linked gastrointestinal symptoms and altered gut microbiota in Covid-19 during and after the infection. On SARS-CoV-2 infection, growing evidence supports the role of gut microbiota in influencing Covid-19 severity and post-Covid effects [2]. Dysbiosis, an imbalance in the gut microbiota composition, is a critical factor in the development of various diseases. Severe Covid-19 cases have been associated with alteration of the intestinal microbiota that may persist for up to a year following the initial infection [3, 4]. However, until now, it was known that Covid-19 can alter the composition of the intestinal microbiota, but we were unaware of the causal effects that the post-Covid microbiota can have on the host’s physiology.
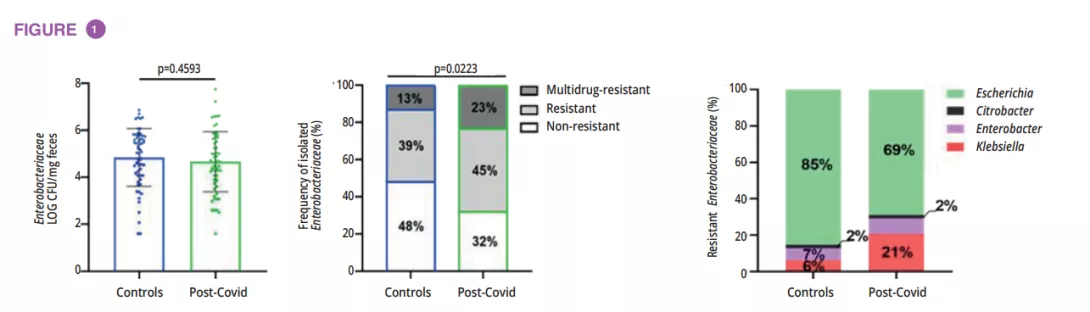
Microbiota analysis of 72 individuals with a history of Covid-19 (post-Covid group) and 59 healthy controls showed no significant differences in gut microbiota diversity (α and β diversity) between the groups, while post-Covid subjects exhibited a higher prevalence of Enterobacteriaceae strains with drug-resistant phenotypes. A higher proportion of post-Covid individuals reported antibiotic use, likely due to Covid-19 treatment. Importantly, Klebsiella strains, associated with antimicrobial resistance (AMR), were notably increased in post-Covid gut microbiota (figure 1).
To understand the direct contribution of post-Covid microbiota to the host, fecal microbiota transplantation (FMT) was performed in germ-free mice using samples from post-Covid and control donors. Post-Covid mice exhibited lung inflammation (figure 2A).
They were also more susceptible to infection with multidrug-resistant Klebsiella pneumonia displaying a more severe lung pathology and inflammatory cell infiltration but were less efficient at clearing the bacteria. Increased Enterobacteriaceae levels in the blood of post-Covid mice suggested systemic translocation. In addition, reduced serum acetate levels were observed in post-Covid Klebsiella pneumonia-infected mice (figure 2A).
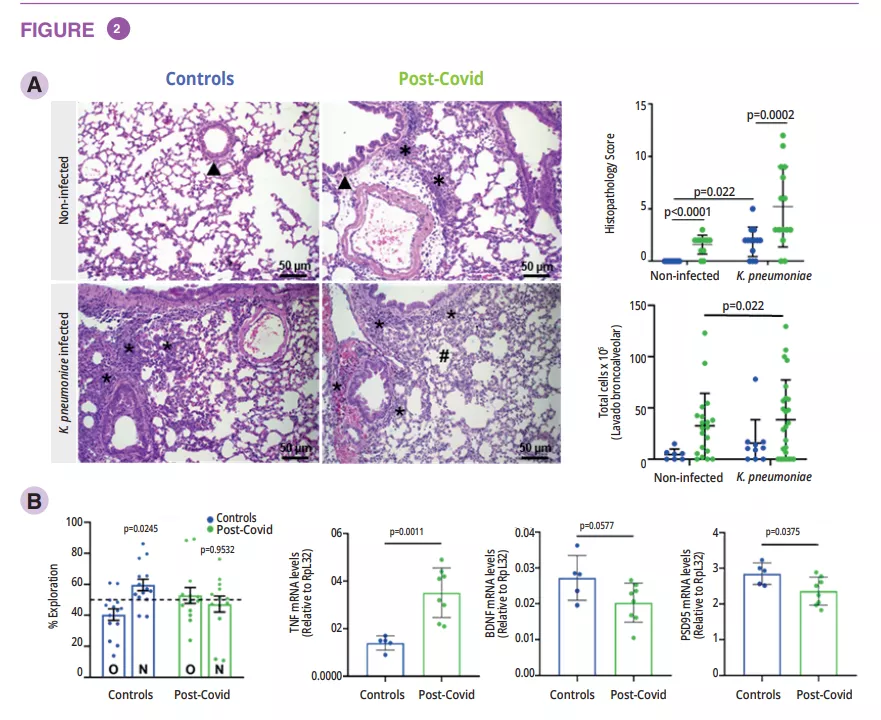
Post-Covid mice exhibited memory impairment in cognitive behavioral tests, along with increased TNF expression and decreased neuroprotective factors in the hippocampus (figure 2B). Administration of a strain probiotic to mice infected with a murine coronavirus prevented memory impairment, reduced weight loss and lung tissue inflammation.
This study warns about the relationship between Covid-19 and the global burden of antimicrobial resistance. Furthermore, it highlights for the first time the causal effect of post-Covid microbiota on lung and nervous system alterations.
The study provides compelling evidence that gut microbiota from individuals following SARS-CoV-2 infection, even after viral clearance, can lead to lung inflammation, cognitive impairment, and increased susceptibility to secondary infections in mice. It highlights the potential for microbiome-based interventions, such as probiotics, to mitigate post-Covid sequelae.
1. Mendes de Almeida V, Engel DF, Ricci MF, et al. Gut microbiota from patients with Covid-19 cause alterations in mice that resemble post-Covid symptoms. Gut Microbes 2023; 15: 2249146.
2. Zuo T, Liu Q, Zhang F, et al. Depicting SARS-CoV-2 faecal viral activity in association with gut microbiota composition in patients with Covid-19. Gut 2021; 70: 276-84.
3. Chen Y, Gu S, Chen Y, et al. Six-month follow-up of gut microbiota richness in patients with Covid-19. Gut 2022; 71: 222-5.
4. Liu Q, Mak JWY, et al. Gut microbiota dynamics in a prospective cohort of patients with postacute Covid-19 syndrome. Gut 2022; 71: 544-52.
COMMENTED ARTICLE Children’s section
By Pr. Emmanuel Mas
Gastroenterology and Nutrition Department, Children’s Hospital, Toulouse, France

The gut microbiome is increasing recognised as playing a role in anorexia nervosa (AN). Studies have reported that AN patients present with dysbiosis compared to healthy controls. However, the underlying mechanisms are unclear and data on influencing factors and the longitudinal impact of microbiome alterations are rare. In this article, the authors presented longitudinal data from 57 hospitalised adolescents diagnosed with anorexia at nine different time points (including a one-year follow-up examination) and compared them to six different time points in 34 healthy controls. The study concluded that characterising prognostically relevant taxa could help stratify patients at admission and potentially identify candidate taxa for future supplementation studies to improve the treatment of anorexia nervosa.
Anorexia nervosa (AN) is a very common psychiatric condition in adolescence, with a high mortality rate. AN is characterised by dysmorphia, reduced calorie intake and malnutrition. Although the pathophysiology of AN is poorly understood, the gut microbiome (GM) is thought to play an important role. GM is actually involved in the gut-brain axis, in malnutrition and also in excess weight, and is altered by diet. The aim of the study was to analyse GM alterations over time in AN patients. It was a one-year study conducted on inpatients until they were discharged from hospital, with an assessment of the clinical parameters associated with the GM in AN.
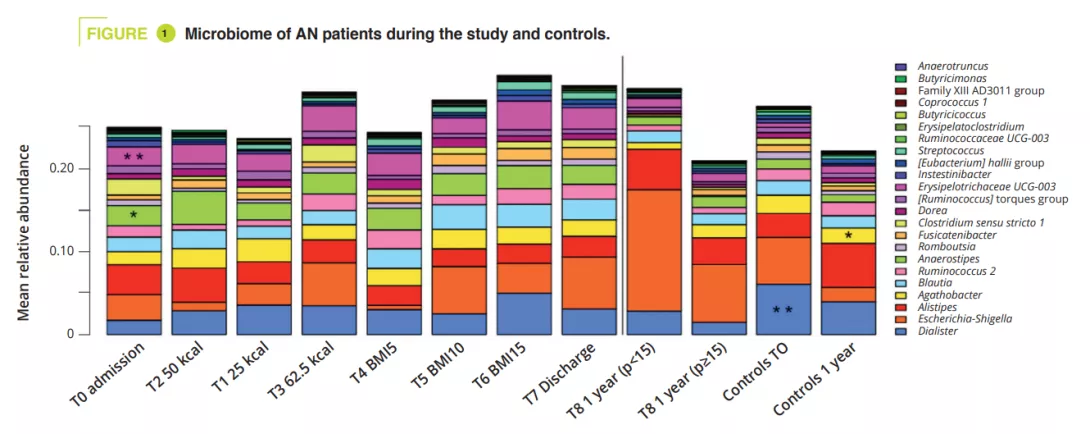
This is the first longitudinal study on gut microbiome (GM) alterations in AN patients, conducted over such a lengthy time frame (one year). The study included 56 patients aged between 12-20 years and 34 controls. Stools were collected at admission and discharge (T0-T7) then one year after admission (T8). Eight patients were re-admitted during the study; patients were separated into those who had recovered their weight (BMI≥15th p [percentile]) and those who still had a low weight (BMI<15th p).
GM composition differed significantly at admission during the acute malnutrition phase, with no difference in terms of alpha-diversity (figure 1). GM differences observed in AN patients compared to controls, even when non-significant, persisted throughout the study. In adolescents with a BMI<15th p at one year, alpha-diversity (Chao1 index) was significantly reduced during hospitalisation compared to admission, discharge and at the 1-year follow-up. A similar trend was observed in AN patients who recovered a BMI≥15 compared to the controls. At admission, the PERMANOVA analysis showed a significant reduction in the genera Legionella, Dialister, Ruminococcaceae UCG-003 and Limnobacter compared to the controls. During in-hospital treatment, the differences between AN patients and controls were reduced, and only remained in the amplicon sequences variants (ASVs). At one year, significant differences were still observed between AN patients with a BMI<15th p and controls in terms of the phyla, classes and orders (p = 0.001 to <0.001), whereas smaller differences were observed between AN patients with a BMI≥15th p and controls (p = 0.063 in terms of ASVs) (figure 2).
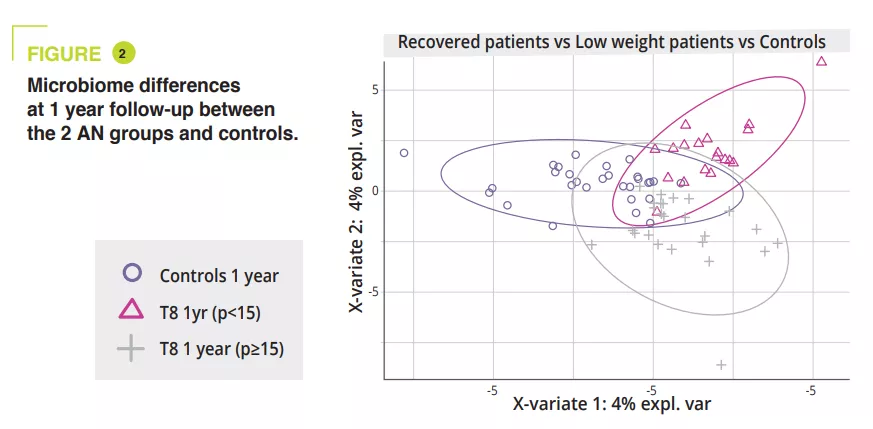
Between admission and the 1-year follow-up, AN patients with a BMI<15th p had a significant abundance of the genera Anaerostipes, Clostridium sensu stricto 1 and Romboustia (p = 0.02) while surprisingly, the GM of AN patients who recovered a BMI≥15th p was more similar during the follow-up. The same was true for changes in GM between hospital discharge and the 1-year follow-up: with a four fold greater abundance of the genus Escherichia-Shigella (p = 0.04) and two fold greater abundance of Alistipes (p = 0.03) in AN patients with a BMI<15th p. GM analysis at admission revealed a significant association between illness duration (phylum-family level, p = 0.011 to 0.022) and amount of weight loss (class-genera level, p = 0.030 to 0.047). A longitudinal PERMANOVA analysis, with correction for the use of laxatives, showed a significant association between GM and the amount of ingested calories (p = 0.003, R2 = 0.009), the BMI-SDS (p = 0.006, R2 = 0.008) and leptin concentration at admission, discharge, and 1-year follow-up (p = 0.02, R2 = 0.02). The genera Ruminiclostridium 5 (p=0.006) and Intestinibacter (p= 0.03) were associated with the risk of hospital readmission. A linear model analysis, with correction for laxative use, illness duration, weight loss and BMI-SMS at admission, identified that at admission four genera were associated with BMI-SDS at the 1-year follow-up: Sutterella, Parasutturella, Lachnospiraceae FCS020 group and Clostridium stricto sensu (p = 0.008 to 0.04) (figure 3).
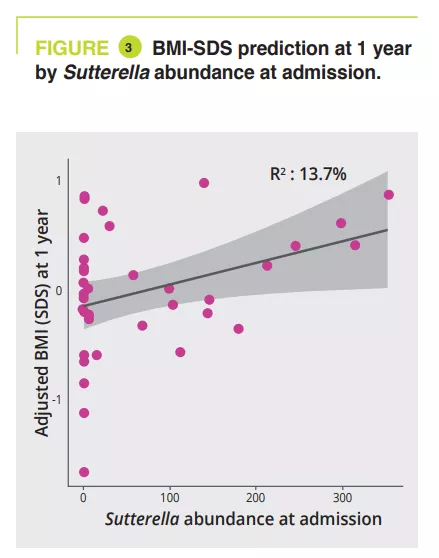
Dysbiosis is observed in acute-phase AN patients and improves partly with treatment. GM composition at admission can help predict the risk of relapse in the first year and improvement in BMI at one year. Thus, a GM analysis at admission could identify the genera and taxa Parasutturella, Lachnospiraceae FCS020 group, Clostridium stricto sensu and uncultured Alistipes as indicative of a poorer prognosis. As a higher abundance of Sutterella is indicative of a positive outcome, it could be used as a probiotic target.
This study showed that GM composition was associated with the duration of the AN and weight loss at admission, but also that GM alterations during treatment was influenced by the calories ingested, weight gain and leptin.
COMMENTED ARTICLE - Adults’ section
By Pr. Harry Sokol
Gastroenterology and Nutrition Department, Saint-Antoine Hospital, Paris, France
Faecal microbiota transplantation (FMT) represents a potential strategy to overcome resistance to immune checkpoint inhibitors in patients with refractory melanoma; however, the role of FMT in first-line treatment settings has not been evaluated. The authors conducted a multicentre Phase I trial combining healthy donor FMT with the PD-1 inhibitors nivolumab or pembrolizumab in 20 previously untreated patients with advanced melanoma. Safety was the primary endpoint. No grade 3 events were reported during the FMT. Five patients (25%) experienced grade 3 immunerelated adverse events from the combination therapy. Key secondary endpoints were objective response rate, changes in gutmicrobiome composition and systemic immune and metabolomics analyses. The objective response rate was 65% (13 out of 20), including four (20%) complete responses. Longitudinal microbiome profiling revealed that all patients engrafted strains from their respective donors. However, the acquired similarity between donor and patient microbiomes was only increased over time in responders. Responders experienced an enrichment of immunogenic bacteria and a loss of deleterious bacteria after FMT. The results showed that FMT from healthy donors is safe in a first-line setting and warrants further investigation when used in combination with immune checkpoint inhibitors.
Almost half of patients with advanced melanoma receiving anti-PD-1 monotherapy develop primary resistance, highlighting the need to develop new therapeutic strategies to improve the response to immune checkpoint inhibitors (ICIs). Although the combination of anti-PD-1 and anti-CTLA4 (cytotoxic T lymphocyte-associated antigen-4) increases the response rate, this therapy is limited by the high number of immune-related adverse events (IR-AEs). The gut microbiome has emerged as an essential regulator of local and systemic immune responses. Several studies in cancer patients treated with ICIs have shown that specific gut bacteria are associated with both immune system response and adverse events [1]. More specifically, the presence of certain commensal genera, such as Ruminococcus, Faecalibacterium and Eubacterium, has been associated with positive outcomes in melanoma patients [2]. The therapeutic potential of the gut microbiome was first demonstrated in mouse models combining ICIs with FMT using faeces from non-responder (NR) patients who were associated with ICI resistance [1]. Two studies showed that FMT in patients with a long-term response to ICI therapy circumvented anti-PD-1 resistance in almost 30% of patients with ICI–refractory melanoma [3, 4]. In these studies, the microbiota of patients changed after FMT, and an increase in Ruminococcaceae and Bifidobacteriaceae was observed in responder (R) patients plus a reprogramming of the tumour microenvironment with increased CD8+ T-cell infiltration and interferon-γ signalling. These clinical findings confirm the potential of microbiome-based interventions to overcome ICI resistance in melanoma.
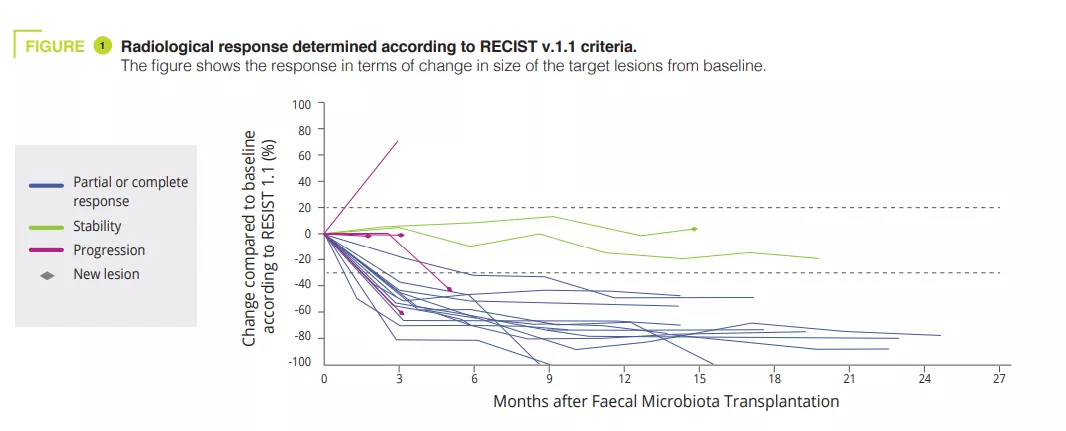
In this article, the authors reported the clinical and translational findings from a Phase I trial (NCT03772899) combining FMT from healthy donors with the PD-1 inhibitors nivolumab or pembrolizumab in treatment-naive patients with advanced melanoma (figure 1). The toxicity observed (85% IR-AEs, of which 25% grade 3 toxicity and zero grade 4 or 5 toxicity) was similar to that reported in the Phase III trials for anti-PD-1. The observed clinical efficacy (objective response 65%) was higher to that of nivolumab and pembrolizumab monotherapy in Phase III trials (objective response 42-45%) and in real-world data (objective response 17.2-51.6%). However, the absence of a control arm and the small size of the study hindered the interpretation of the results.
Unlike the previous studies [3, 4], it included patients receiving first-line treatment, a single FMT was performed by oral capsule, donors were healthy subjects (and not ICI responders) and, finally, only PEG (without the use of antibiotics) was used for the preparation. By studying the microbiota of donors and recipients, the authors observed that the microbiota of responders was enriched in Ruminococcus SGB15234 and SGB15229, Alistipes communis, Eubacterium ramuleus and Faecalibacterium SGB15346, while the abundance of Enterocloster aldensis and Enterocloster clostridioformis decreased. In previous studies, the increase in Faecalibacterium was also associated with the response to ICI [3, 4].
The authors then experimented on mice colonised with human microbiota and observed a similar efficacy of the faecal transplantation from healthy subjects in this context, with an effect associated with an increase in the infiltration of CD8+ T memory lymphocytes in the tumour microenvironment.
Despite its limitations, this study suggested that microbiota modulation via FMT could increase ICI efficacy when administered in a first-line setting for metastatic melanoma. Although the wide-scale use of FMT seems difficult in current practice, modulating the microbiota, in particular with new-generation probiotics, in combination with ICI could become a standard treatment.
This study has shown that FMT from healthy donors is feasible and safe in patients treated with ICI in a first-line setting for metastatic melanoma. Modulating the microbiota through FMT or other methods could increase ICI efficacy although larger controlled studies are required to confirm the data.
1. Routy B, Lenehan JG, Miller WH Jr, et al. Fecal microbiota transplantation plus anti-PD-1 immunotherapy in advanced melanoma: a phase I trial. Nat Med 2023; 29: 2121-32.
2. Gopalakrishnan V, Spencer CN, Nezi L, et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 2018 ; 359 : 97–103.
3. Baruch EN, Youngster I, Ben-Betzalel G, et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science 2021 ; 371 : 602–9.
4. Davar D, Dzutsev AK, McCulloch JA, et al. Fecal microbiota transplant overcomes resistance to anti-PD-1 therapy in melanoma patients. Science 2021 ; 371 : 595–602.
By Dr. Sanda Maria Cretoiu
Department of Morphological Sciences, Cell and Molecular Biology and Histology, “Carol Davila” University of Medicine and Pharmacy Bucharest, Romania

Intestinal disorders can manifest symptoms such as frequent and loose stools, known as diarrhea. This signal from the digestive system can occur for many reasons, from infections and reactions to certain foods to adverse reactions to medications and pre-existing health conditions - summarized in [1]. The intestinal microbiota, i.e., the totality of microorganisms present in the intestine, is essential for the preservation of digestive health and for its impact on the functioning of the intestine. Recent studies illustrate the link between the microbiota and diarrhea of diverse etiology. A balanced and diverse microbiota is vital for overall digestive health, nutrient absorption, and immune system regulation. Currently, there is a tendency towards the large-scale introduction of ways to reprogram the intestinal microbial community: prebiotics, probiotics and postbiotics or the transplantation of fecal matter in order to prevent or treat diarrhea. Research on microbiota modulation will offer actionable strategies for diarrhea prevention and treatment in the near future. The following overview covers the main diarrheal illnesses related to dysbiosis and some aspects regarding microbiota management to ameliorate these gastrointestinal afflictions.

Bacterial, viral or parasitic gut infections cause acute diarrhea and are frequently spread through contaminated water. Most cases of diarrhea are improved in a few days, but severe diarrhea can lead to serious dehydration and can become lethal [9].
Rotaviruses remains the primary cause of diarrhea-associated deaths in children [11], and management of this viral disease generally involves oral or intravenous hydration, tailored to the severity of dehydration [12]. Furthermore, based on the latest conclusions from the ESPGHAN committee (2023) [13], healthcare providers might suggest certain probiotic strains for acute gastroenteric episodes in children, acknowledging their potential (certainty of evidence: low; grade of recommendation: weak) to decrease the duration of diarrhea, and/or hospital stay, and/or volume of fecal discharge. However, a randomized, double-blind, controlled trial of Bolivian children with acute rotavirus diarrhea demonstrated a decreased duration of diarrhea by using an oral rehydration solution plus a mixture of probiotics by comparison with simple rehydration solution [11].
More than 60% of the adults from developed countries who travel to developing countries experience acute diarrhea, also known as traveler’s diarrhea (TD). The most frequently identified pathogens implicated in traveler’s diarrhea episodes are Escherichia coli, Campylobacter jejuni, Salmonella species and Shigella species. Thus, the recommended treatment strategies include antibiotic therapy with azithromycin or fluoroquinolones for moderate to severe cases [14]. However, antibiotics are not recommended to prevent TD, due to insufficient evidence of their prophylactic efficacy and partially due to the risk of antibiotic resistance [15].
There is conflicting data regarding the efficacy of probiotics in preventing traveler’s diarrhea [16]. One systematic review and meta-analysis compared the efficacy of rifaximin and probiotics in preventing TD. [15].
Antibiotics are one of the most prescribed medications and represent an effective treatment for several infectious pathologies [17]. One of the complications associated with antibiotic therapy is antibiotic-associated diarrhea (AAD), which occurs in 5%-35% of the patients who receive antibiotherapy [18]. AAD can be defined as three or more watery or loose stools per day for at least two consecutive days, which is strictly related to antibiotics administration and no other cause [14]. The highest risk is attributed to aminopenicillins, cephalosporins and clindamycin, which primarily target anaerobes [19].
The lack of an infectious agent identified in AAD may be explained by the direct toxic effect of the antibiotics on the intestinal mucosa, which may cause diarrhea. Due to their beneficent properties, probiotics are now being researched and used for both treatment and prophylaxis of AAD [16, 18].
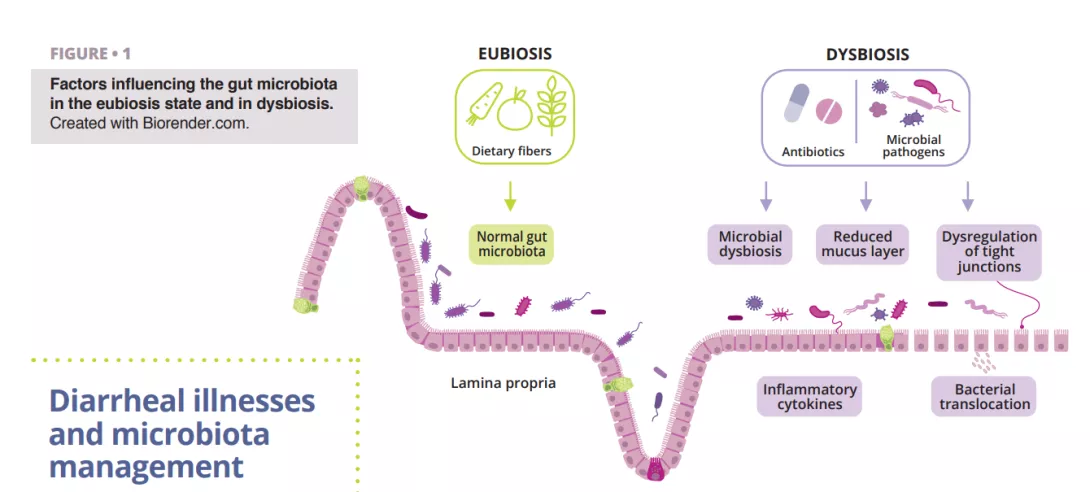
Clostridioides difficile (CD) infection is the most common cause of nosocomial antibiotic-associated diarrhea in adults. Risk factors include age over 65 years, long hospitalization in intensive care, and administering antibiotics (fluoroquinolones, clindamycin, cephalosporins, and betalactams in particular) or proton pump inhibitors. During antibiotherapy, anaerobes that produce SCFAs may disappear due to antibiotic-induced alterations in the gut microbiota, which may also disturb the metabolism of carbohydrates and bile and cause an osmotic imbalance. Following antibiotic intake, all three intestinal barriers are affected: the epithelial intestinal cells, the mucus and antimicrobial peptides layer, and the immunoprotective layer composed of different immune cells and various biomolecules (figure 1). This event can interfere with the production of mucin, cytokines, and antimicrobial peptides, dysregulating intestinal function and leading to other infections or even causing recurrent episodes of infections. The American Gastroenterological Association (AGA) conditionally recommends specific probiotics for preventing CD infection in individuals on antibiotics, noting that the quality of evidence is low [20].
Recent breakthroughs in microbiota research, including metagenomic analysis and microbial transplantation, are revolutionizing our approach to diarrhea treatment (figure 2).
Treatment options for diarrhea should take into account the causative mechanisms involved in the genesis of diarrhea, from infectious toxins capable of disrupting fluid and electrolyte balance to patients who developed dysbiosis due to other causes and patients with large amounts of non-absorbed carbohydrates in the lumen triggering osmotic diarrhea.
There is limited data regarding the prebiotics and fibers in treating diarrhea (table 2). Apparently, prebiotics are more prone to prevent and treat the recurrence of diarrhea. At the same time, fibers, mainly the viscous ones, are more indicated during acute episodes due to their water-retaining capacity. Other therapeutic options involve, in some cases, the probiotic administration and (table 3), in severe cases, the use of fecal microbiota transplantation (FMT).
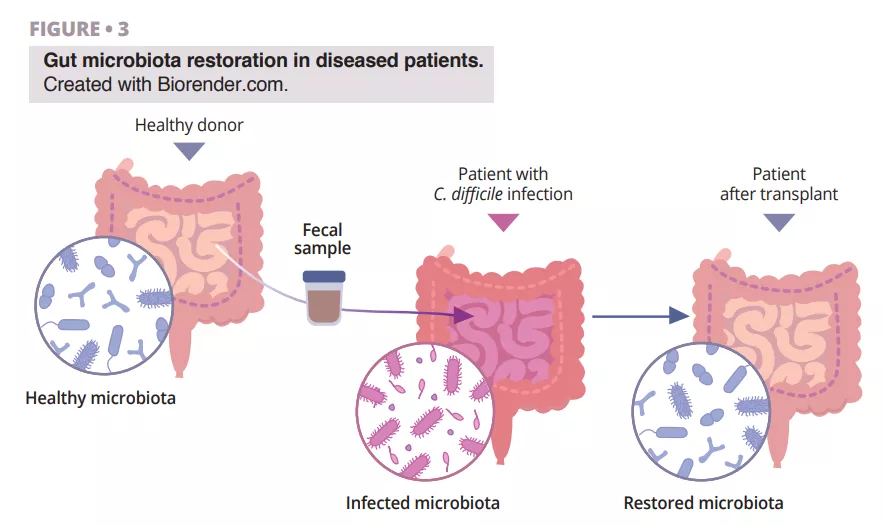
The fascinating journey of FMT discovery has roots in ancient China, where Ge Hong treated patients with severe diarrhea using a “yellow soup” consisting of feces suspension. In modern times, Dr. Ben Eiseman used fecal enemas from healthy individuals to treat pseudomembranous enterocolitis back in 1958. Nowadays, there is growing interest in fecal microbiota transplantation (FMT) as a treatment for recurrent Clostridioides difficile infection (CDI), which points out its utility [22]. Research is ongoing regarding its efficacy towards inflammatory bowel disease, diabetes, cancer, liver cirrhosis, and brain diseases such as Parkinson’s [23]. The benefits of using FMT in patients with diarrhea are based on the idea that the healthy microbial flora introduced via FMT has the ability to outcompete pathogens and restore the composition of a healthy gut microbiome (figure 3).


Research reveals that reduced gut microbiota diversity is associated with increased susceptibility to diarrhea, paving the way for potential diagnostic and therapeutic interventions. Maintaining a balanced and diverse gut microbiota prevents diarrhea and promotes overall digestive health. Imbalances in the microbiota, known as dysbiosis, can result from infectious acute diarrhea or dysbiosis due to other factors (frequent antibiotic use, unhealthy diet, malabsorption) that can contribute to chronic diarrhea. Understanding the complex interplay between microbial composition and clinical symptoms is crucial for personalized patient management of diarrhea. Tailored approaches based on unique microbiota profiles can lead to more effective strategies or interventions. The introduction of probiotics and a diet rich in prebiotics, microbiota transplantation, integration of multi-omics approaches, innovative use of machine learning, and the growing trend of interdisciplinary research collaborations may help restore microbial balance and support gastrointestinal well-being. Hopefully, in the future, one could design microbiome-based therapies as suggested by Peter J. Turnbaugh, laying the base for new treatment principles [25].
1. Iancu MA, Profir M, Roşu OA, et al. Revisiting the Intestinal Microbiome and Its Role in Diarrhea and Constipation. Microorganisms 2023; 11: 2177.
2. David L, Weil A, Ryan ET, et al. Gut microbial succession follows acute secretory diarrhea in humans. mBio 2015; 6: 1-14.
3. Sohail MU, Al Khatib HA, Al Thani AA, et al. Microbiome profiling of rotavirus infected children suffering from acute gastroenteritis. Gut Pathog 2021; 13: 21.
4. Becker-Dreps S, Allali I, Monteagudo A, et al. Gut Microbiome Composition in Young Nicaraguan Children During Diarrhea Episodes and Recovery. Am J Trop Med Hyg 2015; 93: 1187-93.
5. Cannon JL, Seabolt MH, Xu R, et al. Gut Microbiome Changes Occurring with Norovirus Infection and Recovery in Infants Enrolled in a Longitudinal Birth Cohort in Leon, Nicaragua. Viruses 2022; 14: 1395.
6. Azagra-Boronat I, Massot-Cladera M, Knipping K, et al. Strain-Specific Probiotic Properties of Bifidobacteria and Lactobacilli for the Prevention of Diarrhea Caused by Rotavirus in a Preclinical Model. Nutrients 2020; 12: 498.
7. Kim AH, Lee Y, Kim E, et al. Assessment of oral vancomycin-induced alterations in gut bacterial microbiota and metabolome of healthy men. Front Cell Infect Microbiol 2021; 11: 629438.
8. Binder HJ. Role of colonic short-chain fatty acid transport in diarrhea. Annu Rev Physiol 2010; 72: 297-313.
9. Collinson S, Deans A, Padua-Zamora A, et al. Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst Rev 2020; 12:CD003048.
10. Desselberger U. Viral gastroenteritis. Medicine 2017; 45: 690-4.
11. GBD 2016 Diarrheal Disease Collaborators. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of diarrhea in 195 countries: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect Dis 2018; 18: 1211-8.
12. Iturriza-Gómara M, Cunliffe NA. 34 - Viral Gastroenteritis. Ryan ET, Hill DR, Solomon T, Aronson NE, Endy TP. (eds) Hunter’s Tropical Medicine and Emerging Infectious Diseases (tenth edition). Elsevier, 2020, pp. 289-307.
13. Szajewska H, Berni Canani R, Domellöf M et al.; ESPGHAN Special Interest Group on Gut Microbiota and Modifications. Probiotics for the Management of Pediatric Gastrointestinal Disorders: Position Paper of the ESPGHAN Special Interest Group on Gut Microbiota and Modifications. J Pediatr Gastroenterol Nutr 2023; 76: 232-47.
14. Kopacz K, Phadtare S. Probiotics for the Prevention of antibiotic-associated diarrhea. Healthcare 2022; 10: 1450.
15. Fan H, Gao L, Yin Z, et al. Probiotics and rifaximin for the prevention of travelers’ diarrhea: A systematic review and network meta-analysis. Medicine 2022; 101: e30921.
16. Girardin M, Seidman EG. Indications for the use of probiotics in gastrointestinal diseases. Dig Dis 2011; 29: 574-87.
17. Goodman C, Keating G, Georgousopoulou E, et al. Probiotics for the prevention of antibiotic-associated diarrhoea: a systematic review and meta-analysis. BMJ Open 2021; 11: e043054.
18. McFarland LV. Antibiotic-associated diarrhea: epidemiology, trends and treatment. Future Microbiol 2008; 3: 563-78.
19. Barbut F, Meynard JL. Managing antibiotic associated diarrhoea. BMJ 2002; 324: 1345-6.
20. Su GL, Ko CW, Bercik P, et al. AGA Clinical Practice Guidelines on the Role of Probiotics in the Management of Gastrointestinal Disorders. Gastroenterology 2020; 159: 697-705.
21. Codex AC. Report of the 31th session of the codex committee on nutrition and foods for special dietary uses. Rome, Italy: FAO/WHO 2009.
22. Peery AF, Kelly CR, Kao D, et al.; AGA Clinical Guidelines Committee. AGA Clinical Practice Guideline on Fecal Microbiota-Based Therapies for Select Gastrointestinal Diseases. Gastroenterology 2024; 166: 409-34.
23. Tariq R, Disbrow MB, Dibaise JK, etal. Efficacy of Fecal Microbiota Transplantation for Recurrent C. Difficile Infection in Inflammatory Bowel Disease. Inflamm Bowel Dis 2020; 26: 1415-20.
24. Guarino A, Ashkenazi S, Gendrel D, et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition/European Society for Pediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe: update 2014. J Pediatr Gastroenterol Nutr 2014; 59: 132-52.
25. Rock RR, Turnbaugh PJ. Forging the microbiome to help us live long and prosper. PLoS Biol 2023; 21: e3002087.
A study explains how certain vaginal bacteria can alter the cervicovaginal environment and have a negative impact on reproduction. How they do it: protein transport vesicles.
One of the features of the vaginal microbiota is that it is balanced when it is largely dominated by Lactobacillus crispatus. On the other hand, colonization by anaerobic bacteria such as Gardnerella vaginalis and Mobiluncus mulieris is associated with an increased risk of STIs, bacterial vaginosis and preterm birth. However, the mechanisms involved remain unclear, although the literature suggests that the production of extracellular vesicles by certain bacteria is involved. Could this mechanism be at work in the reproductive system? This is the hypothesis 1 put forward and validated by the team led by Dr. Michal A. Elovitz of the Icahn School of Medicine at Mount Sinai 2 in New York, who studied, in vitro, the extracellular vesicles produced by the beneficial L. crispatus and the detrimental G. vaginalis and M. mulieris.
After using electron microscopy to observe the actual presence of vesicles ranging from 90 to 420 nm in diameter in cell culture media, the team analyzed their contents. The vesicles produced by G. vaginalis, M. mulieris and L. crispatus contained 491, 336 and 247 bacterial proteins, respectively. Several of these were of functional interest: the G. vaginalis cargo was rich in vaginolysin, a toxin capable of inducing cell lysis (breakdown of the cell wall) in cervicovaginal epithelial cells and common in bacterial vaginosis; that of M. mulieris contained proteins capable of stimulating an immune response, while several proteins in L. crispatus vesicles protect the epithelial barrier.
What's more, these bacterial vesicles rapidly internalized (1-4 hours) into cervical and vaginal epithelial cells. All that remained was to determine their effects.
Exposure of cervical and vaginal epithelial cells to the contents of G. vaginalis and M. mulieris bacterial vesicles induced a dose-dependent immune response. The response of endocervical cells was more pronounced than that of ectocervical cells. In contrast, L. crispatus did not induce any significant cytokine overexpression.
Thus, cervical and vaginal epithelial cells respond with overproduction of cytokines when exposed to G. vaginalis and M. mulieris vesicles, but not to L. crispatus. This immune response is mediated by signaling pathways activated by the (sidenote: TLR2 receptor Toll-like receptor (molecular pattern recognition) located in the cell membrane, encoded by the TLR2 gene and involved in the recognition of various pathogens, including bacteria, viruses, fungi and parasites. Source: Oliveira-Nascimento L, Massari P, Wetzler LM. The Role of TLR2 in Infection and Immunity. Front Immunol. 2012 Apr 18;3:79. ) .
Bacteria thus have an effect, whether detrimental or beneficial, on reproductive health via bacterial extracellular vesicles: these act as couriers, providing protection for bacterial proteins during transport, then delivering them to epithelial cells and ultimately inducing a host immune response.
 Bacterial vaginosis: sexual transmission & genomic insights
Bacterial vaginosis: sexual transmission & genomic insights
 Menstrual toxic shock syndrome: balanced flora protects against attacks by S. aureus
Menstrual toxic shock syndrome: balanced flora protects against attacks by S. aureus
A study has just shown that resistant starch induces changes in the structure of gut microbiota that are conducive to weight loss and beneficial for health. Supplementation with 40 g/day enabled volunteers to lose more than 6 pounds in 2 months, without dieting!
The gut microbiota
This could be the umpteenth "Spring Special" issue of a women's magazine extolling the virtues of the latest fad diet...But no, these are the results of a particularly rigorous study published in the journal Nature Metabolism 1 that is generating interest and presenting the potential weight-loss benefits of resistant starch. By modulating the structure of our gut microbiota, this dietary fiber—found in legumes, whole grains and green bananas—could well help us shed unwanted pounds and improve our health. Let’s take a closer look at the results.
The researchers recruited 22 overweight men and 15 overweight women ( (sidenote: Body Mass Index (BMI) Body Mass Index (BMI) assesses the corpulence of an individual by estimating the body fat mass calculated by a ratio between weight ((kg) and height squared (m2). https://www.nhlbi.nih.gov/health/educational/lose_wt/BMI/bmicalc.htm https://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi ) > 24) with a normal (non-hypocaloric) balanced diet, and supplemented them with either resistant starch or "normal" placebo starch (see box). They measured changes in BMI, gut microbiota and various metabolic parameters.
The results first indicate that taking 40 g of starch daily for 2 months is associated with an average weight loss of 6.17 lbs, with in particular:
None of these effects were observed with the placebo starch.
This study is a "randomized, double-blind, placebo-controlled crossover trial," one of the most rigorous scientific methods in medical research. Participants were randomly divided into two groups:
In order to eliminate any human factor that might influence the results, neither the researchers nor the participants knew the type of starch being given.
Analyses indicate that resistant starch led to a veritable remodeling of the structure of the volunteers' gut microbiota, with an increase in several bacterial strains, particularly Bifidobacterium adolescentis. The researchers' calculations show that the abundance of this bacteria is strongly correlated with the reduction in BMI and visceral fat.
When they transplanted the fecal microbiota of volunteers who had consumed resistant starch into mice (sidenote: Fecal Microbiota Transplantation (FMT) A therapeutic procedure to restore the gut microbiota by transferring fecal bacteria from a healthy donor to a recipient. Explore https://www.science.org/doi/10.1126/scitranslmed.abo2750 ) , the researchers found the same type of effects on weight and insulin sensitivity. And the same effects were obtained by supplementing mice with the beneficial bacteria identified as B. adolescentis.
This reinforces the hypothesis that the changes in the gut microbiota induced by resistant starch are responsible for its beneficial effects.
Starch is the form in which plants (rice, potatoes, wheat, corn, etc.) store carbohydrates. Resistant starch is a part of the starch that is not broken down by our digestive enzymes, and therefore arrives intact in the colon. It is fermented by bacteria in the microbiota, which in turn release various beneficial substances, including short-chain fatty acids
(sidenote:
Short chain fatty acids (SCFA)
Short chain fatty acids (SCFA) are a source of energy (fuel) for an individual’s cells. They interact with the immune system and are involved in communication between the intestine and the brain.
Silva YP, Bernardi A, Frozza RL. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front Endocrinol (Lausanne). 2020;11:25.
)
. 2 Oats, rice, sorghum, barley, beans and legumes, as well as green bananas, provide large quantities of fiber and resistant starch. 3
Cooking reduces the resistant starch content of foods, but cooling (e.g. rice or potato salad) helps to restore it. 2
The changes in the gut microbiota, among other things:
These results need to be confirmed by a larger-scale study. But they suggest that increasing the resistant starch content of one's diet is both a simple and powerful strategy for losing weight...or keeping it off after dieting!
Behind this strange-sounding name lies a very common digestive disorder which involves pain and/or discomfort centered on the stomach. One effective and pertinent solution seems to be modulating the gut microbiota with probiotics.
The gut microbiota
Functional dyspepsia: a strange-sounding name for a condition you’re surely familiar with, since it affects almost 1 in 10 people, particularly women, smokers, and patients taking non-steroidal anti-inflammatory drugs (ibuprofen, ketoprofen). Functional dyspepsia is a common digestive disorder characterized by chronic pain or discomfort centered on the stomach.
Common symptoms are an unpleasant feeling of being too full after a normal-sized meal, of feeling full when you’ve just started eating, or of pain and/or burning at the bottom of the stomach. All this can last for weeks on end.
250-400 m2 the human digestive system is the largest exchange surface in the body.
98% Proteobacteria, Firmicutes, Actinobacteria, and Bacteroidetes make up over 98% of the total gut microbiota in healthy individuals.
While the many causes and mechanisms of functional dyspepsia are still poorly understood, the gut microbiota has been singled out for blame. For example, the Helicobacter pylori bacterium that sometimes makes itself at home in the stomach appears to be a major contributor to the development and progression of the disease, probably via inflammation of the gastrointestinal mucosa and disruption of gut motility.
40% Over 40% of patients worldwide suffer from functional gastrointestinal disorders, of which functional dyspepsia is one of the most common.
3% to 5% Functional dyspepsia accounts for 3% to 5% of visits to family doctors.
In more general terms, the gut microbiota as a whole may be involved. This may be both directly (an imbalance in composition and abundance may lead to gut dysfunction) and via the bacterial metabolites it produces, which can have a protective (e.g. (sidenote: Short chain fatty acids (SCFA) Short chain fatty acids (SCFA) are a source of energy (fuel) for an individual’s cells. They interact with the immune system and are involved in communication between the intestine and the brain. Silva YP, Bernardi A, Frozza RL. The Role of Short-Chain Fatty Acids From Gut Microbiota in Gut-Brain Communication. Front Endocrinol (Lausanne). 2020;11:25. ) produced by Firmicutes) or harmful (pro-inflammatory sphingolipids produced by Bacteroidetes and Prevotellaceae) effect on the host.
2/3 of those affected by functional dyspepsia suffer from persistent, irregular symptoms that can affect their quality of life and even their psychological well-being.
8 times Patients with functional dyspepsia have an approximately eight-fold higher risk of irritable bowel syndrome than the general population.
One direct consequence of the gastrointestinal microbiota’s involvement in functional dyspepsia is the search for solutions to restore this microbiota, and thus host’s (patient’s) health. Several trials 1 on patients appear to show that probiotics can significantly improve symptoms by restoring the balance of the gut flora.
Another, slightly more aggressive, approach is the use of broad-spectrum antibiotics which eliminate undesirable bacteria. This nuclear option is highly effective, particularly in women, but causes considerable collateral damage to good bacteria. Hence the need for further research to better understand the mechanisms involved and propose targeted, effective diagnostic methods and treatments.



